|
MEMBERSHIPS WILL BE GOOD THROUGH 2018! GSGA membership runs on a calendar year basis so you will need to renew your membership each year. You can do that in a couple of different ways: • Visit the pro shop at your home club to renew. • to renew online at a participating online membership club. When renewing your GSGA membership online, be sure to enter your existing GHIN number in the application form or you will be assigned a new number. If you have resigned your club membership but want to continue to maintain your handicap and GSGA membership. • You may transfer your membership to your new club (contact the pro shop). • and join one of our online membership clubs and transfer your GHIN number and handicap records there. The cost to renew online is $40. To learn more about the benefits of GSGA membership. You may also click the link below for answers to frequently-asked questions. GHIN: Golf Handicap and Information Network Username: Password. Jun 25, 2014 Re-establishing a handicap. By Chris Stewart, June 24, 2014 in Golf Talk. If you had GHIN, your old scores will pop up (probably after a revision.). Welcome to the GHIN Handicap Program (GHP) Online Club. In the Handicap Index. You are adding a golfer to your club that does not have an existing GHIN. Be sure to search and download the MGA HOME mobile app. Hold an active MGA/USGA GHIN Handicap Index. Club in your area and ask them to activate you. Welcome to the GHIN Handicap Program. Activate or inactivate a golfer or multiple. After you have entered the GHIN number of the person to add to your.  
GOLF TOURNAMENT DETAILS • What: Phoenix Premier Golf Club 2-Person Shamble Tournament • Where: Lone Tree Golf Club • When: Saturday, December 6, 2014 • Start Time: 9:00 AM • Event Format: 2-person Shamble • Tee Format: Shotgun • Entry Fee: $65.00 – Premier Golf Club Members; $75.00 – Guests • ***REGISTRATION CLOSED*** • Fee Includes: Green Fee, Cart, Tax, Range Balls! • Additional Notes: There is an optional $40 prize fund buy-in and $40 flighted skins game for this event. BOTH THE PRIZE FUND AND SKINS GAME will be collected at check-in (cash only). By contributing to the prize fund, you will become eligible to win Net/Gross prizes for 1st and 2nd place (by flight) and Closest-to-Pin. Valid USGA Handicap required to participate. Please provide your GHIN # or instructions on how to verify your handicap when you register. If you would like to request to play with a certain player or group, use the “Additional Information – Order Notes” field when you checkout. 
0 Comments
My dad just got the new iphone 4 16gb for verizon but decided he didn't want it so is giving it to me. However when i went to activate it it put his number on (when plugged into itunes) and he really doesn't want it so i'm trying to put my number on instead. Since i couldn't find a way to change it. My dad just got the new iphone 4 16gb for verizon but decided he didn't want it so is giving it to me. However when i went to activate it it put his number on (when plugged into itunes) and he really doesn't want it so i'm trying to put my number on instead. Since i couldn't find a way to change it via itunes i signed into our verizon account, went to my number, and did 'activate a new device', typed in the serial number, and it said 'We cannot activate the phone associated with the Device ID you entered. Nov 03, 2017 How to Activate a Cell Phone. Then chances are you'll need to activate it. The process is a little. Verizon - (800) 922-0204; T-Mobile. Jul 18, 2011 Best Answer: Call Verizon from a different phone. *611 from a Verizon phone or (800) 922-0204 from another device. Keep selecting 0 to talk to someone. 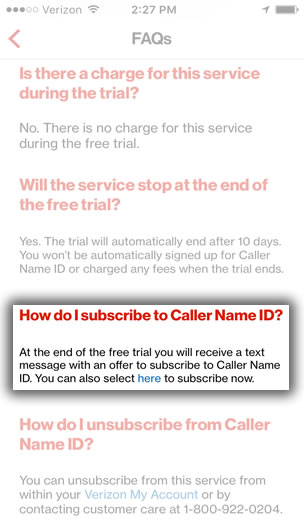
Please contact Customer Service at (800) 922-0204 for assistance.' I have tried calling the *228 like i've always done with new phones but it didn't seem to work and until then i am stuck staring at a nice iphone that i can't use. Someone help? You're able to do it 3 diverse techniques: a million. You could flow as much as the Verizon prompt keep and have them turn on it for you for loose and turn over your previous telephone quantity to your iphone 5. You are going to be able to desire to have wifi for the subsequent decision.you could turn your telephone on with the SIM card in it and then flow for the duration of the step by way of step technique such by way of fact the language, time zone, u. & then after that your telephone ought to be examine for use 3. You could flow on the flow the Verizon prompt internet site and flow to turn on telephone or call 611 out of your telephone and refer to an operator. • Tell us some more • Upload in Progress • Upload failed. Please upload a file larger than 100x100 pixels • We are experiencing some problems, please try again. • You can only upload files of type PNG, JPG, or JPEG. 
• You can only upload files of type 3GP, 3GPP, MP4, MOV, AVI, MPG, MPEG, or RM. • You can only upload photos smaller than 5 MB. • You can only upload videos smaller than 600MB. • You can only upload a photo (png, jpg, jpeg) or a video (3gp, 3gpp, mp4, mov, avi, mpg, mpeg, rm). • You can only upload a photo or a video. • Video should be smaller than 600mb/5 minutes • Photo should be smaller than 5mb •.  
We have a Device Unlocking Policy that covers postpay devices, prepaid devices and the policy for Deployed Military Personnel. Postpay Device Unlocking Policy • We do not lock most phones or tablets that are activated with our postpay service, either during or after the term of your service contract or Device Payment Plan installment sales agreement. • We do not lock our 4G LTE devices, and no code is needed to program them for use with another carrier. • We do not lock our 3G devices, other than our non-iPhone 3G World Devices. The simple code to program such 3G devices for use with another carrier is either “000000” or “123456.” If the user guide for your device does not provide instructions to access the programming menu, you can contact us at *611 from your Verizon Wireless phone, or (800)922-0204, for assistance. • The SIM cards used in our non-iPhone 3G World Devices are locked. We willl unlock those SIM cards at your request. Prepaid Device Unlocking Policy 3G Device Policy • We do not lock our 3G devices, other than our 3G Phone-in-the-Box Prepaid phones, and the simple code to program such 3G Prepaid devices for use with another carrier is either “000000” or “123456.” If the user guide for your device does not provide instructions to access the programming menu, you can contact us at *611 from your Verizon Wireless phone, or (888) 294-6804, for assistance. • Our 3G Prepaid Phone-in-the-Box phones are restricted for use with our Prepaid service for 6 months after activation, and are locked to the Verizon Wireless network for 12 months after activation. After 6 months, the restriction to our Prepaid service is automatically removed. If service is disconnected within the first 6 months, a new 6 month period will begin if the phone is reactivated. After 12 months, you can contact us at *611 from your Verizon Wireless Prepaid phone, or (888)294-6804, and we will provide you with programming instructions so you can unlock your phone. If the user guide for your phone does not provide instructions to access the programming menu, we can help you with that as well. 4G Device Policy • We do not lock our 4G devices, other than our 4G Phone-in-the-Box Prepaid phones. In order to partially offset the subsidy we provide to make our 4G Phone-in-the-Box Prepaid phones more affordable to service customers, our 4G Phone-in-a-Box phones may only be used with Verizon Prepaid service for 1 year from original activation date or until payments in the amount specified on the back of the Phone-in-a-Box package are added to the Prepaid account. • Once either of these criteria is satisfied, you can contact us at *611 from your Verizon Wireless Prepaid phone, or (888) 294-6804, and we will provide you with programming instructions so you can unlock your phone. If the user guide for your phone does not provide instructions to access the programming menu, we can help you with that as well. Unlocking policy for Deployed Military Personnel If you are a Verizon Wireless customer in the military we will unlock your device at your request. Postpay devices • We do not lock our 4G LTE devices, and no code is needed to program them for use with another carrier. • We do not lock our 3G devices, other than our non-iPhone3G World Devices, and the simple code to program such 3G devices for use with another carrier is either “000000” or “123456.” If the user guide for your device does not provide instructions to access the programming menu, you can contact us at *611 from your Verizon Wireless phone, or (800)922-0204, for assistance. • The SIM cards used in our non-iPhone 3G World Devices are locked. We will unlock the SIM card for you at your request. Prepaid devices • Once activated on our network, we do not lock our 4G LTE Prepaid devices, and no code is needed to program them for use with another carrier. To activate a prepaid device on our network, you must make your first monthly service payment. • We do not lock our 3G Prepaid devices, other than our 3G Phone-in-the-Box Prepaid phones, and the simple code to program such 3G Prepaid devices for use with another carrier is either “000000” or “123456.” If the user guide for your device does not provide instructions to access the programming menu, you can contact us at *611 from your Verizon Wireless phone, or (888)294-6804, for assistance. • Our 3G Prepaid Phone-in-the-Box phones are restricted for use with our Prepaid service for six months after activation, and are locked to the Verizon Wireless network for 12 months after activation. After six months, the restriction to our Prepaid service is automatically removed. After 12 months, you can contact us at *611 from your Verizon Wireless Prepaid phone, or (888)294-6804, and we will provide you with the programming code so that you can unlock your phone. If the user guide for your phone does not provide instructions to access the programming menu, we can help you with that as well. PLEASE NOTE: Carriers typically use different frequencies and air interface technologies to provide wireless network access. Accordingly, a device that works on one carrier’s network may not be technologically compatible with another carrier’s network. “Unlocking” a device refers only to disabling software that would prevent a consumer from attempting to activate a device designed for one carrier’s network on another carrier’s network, even if that network is technologically compatible. In other words, “unlocking” a device will not necessarily make a device interoperable with other networks – a device designed for one network is not made technologically compatible with another network merely by “unlocking” it. Additionally, unlocking a device may enable some functionality of the device but not all (e.g., an unlocked device may support voice services but not data services when activated on a different network). Ever since Apple first offered Macs with, they've included support for TRIM, a method for the OS to assist an SSD in freeing up space. TRIM Command The TRIM command is issued by the operating system to assist an SSD in cleaning up data in storage blocks that are no longer needed. This helps the write performance of an SSD by keeping more blocks of data free to be written to. It also keeps the SSD from being aggressive in cleaning up after itself and causing wear on the memory chips, leading to early failure. TRIM is supported in and later, but Apple only enables the TRIM command for use with Apple-supplied SSDs. It’s not clear why Apple limited TRIM support this way, but the conventional wisdom is that TRIM implementation is up to the SSD manufacturer, and each SSD manufacturer uses a different TRIM methodology. As such, Apple only wanted to use TRIM on SSDs that it has certified. 
That left those of us who like to upgrade our Macs out in the cold, at least when it came to running performance-enhancing SSDs. Without support for TRIM, there was a possibility that over time, our expensive SSDs would slow down, and we would see a real performance drop in writing to the SSD. Thankfully, there are a few third-party utilities that can enable TRIM for non-Apple-supplied SSDs, including TRIM Enabler, one of my in 2014. These utilities make use of Apple’s built-in TRIM support; they just removed the ability for the OS to check if the SSD is on Apple’s list of approved manufacturers. Should You Use TRIM? Some early-generation SSDs had unusual implementations of the TRIM function that could lead to data corruption. For the most part, these early SSD models were difficult to come across, unless you picked one up from a source that specialized in used products, such as flea markets, swap meets, or eBay. 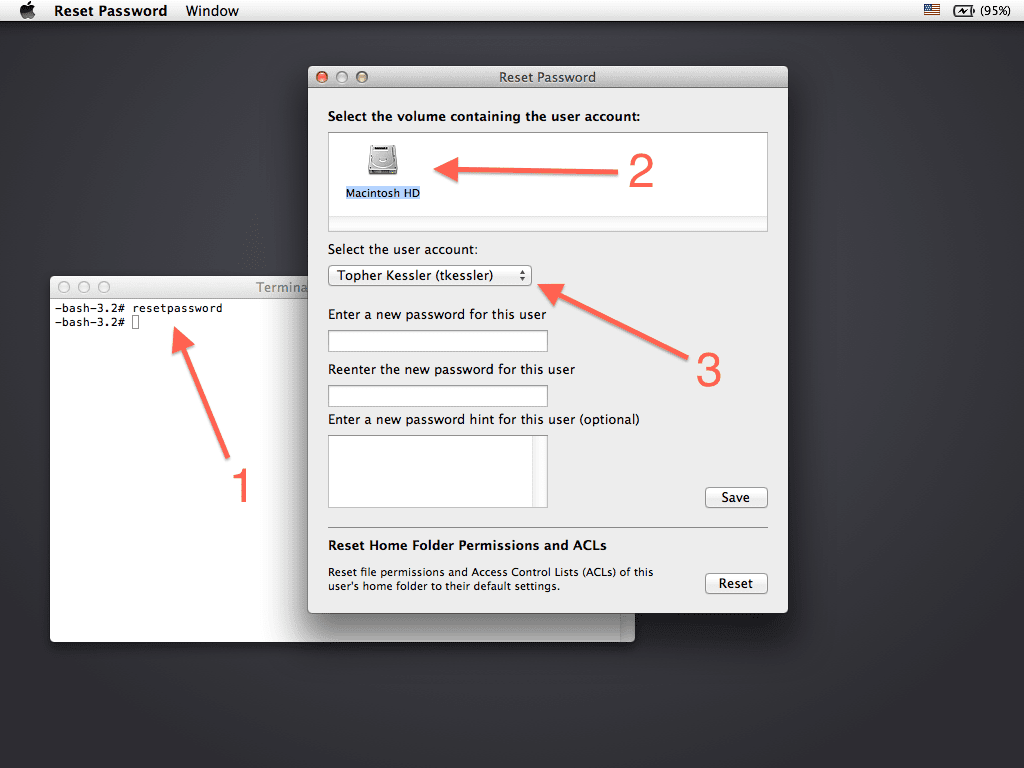
Paragon NTFS for Mac OS X. Won’t be able to use the product until you activate. Downloadable free demo versions and open documentation for all users. Wolfram Mathematica 10.4 Full + Crack is a. Wolfram Mathematica 10.4 Activation. Paragon NTFS 14 Final with Crack Mac OS X Paragon NTFS for Mac OS X 14. TRIM is disabled in OS X for third-party SSDs, but with this simple Terminal command, you can enable TRIM for any SSD you have added to your Mac. One thing you should do is check with the SSD manufacturer to see if there are any firmware updates for the SSD model you have. It’s not just older SSDs that can have problems, though. Some popular SSD models, such as Samsung 840 EVO, 840 EVO Pro, 850 EVO, and 850 EVO Pro, have exhibited problems with TRIM that can lead to data corruption. Luckily for us Mac users, the Samsung TRIM issues seem to only become apparent when used with queued TRIM commands. OS X only makes use of sequential TRIM commands at this time, so enabling TRIM with the Samsung line of SSDs should be OK, as reported by MacNN. The Importance of Backups I’ve been using the TRIM command with the third-party without issues, however, before enabling TRIM I made sure that I had a system in place. Should an SSD exhibit a failure caused by TRIM, it's likely to involve large blocks of data being reset, causing non-recoverable file loss. Always have a in place. How to Enable TRIM in OS X Before you proceed, remember the TRIM function is automatically enabled for Apple-supplied SSDs; you only need to execute the following steps for third-party SSDs you installed as upgrades. • Launch, located in the /Applications/Utilities folder. • At the Terminal command prompt, enter the text below: (Tip: you can triple-click the command line and then copy/paste it into the Terminal window.) sudo TRIMforce enable • When requested, enter your administrator password. • The terminal will then produce one of the more scary warnings that Apple has come up with yet: “IMPORTANT NOTICE: This tool force-enables TRIM for all relevant attached devices, even though such devices may not have been validated for data integrity while using TRIM. Use of this tool to enable TRIM may result in unintended data loss or data corruption. It should not be used in a commercial operating environment or with important data. 
Before using this tool, you should back up all of your data and regularly back up data while TRIM is enabled. This tool is provided on an “as is” basis. APPLE MAKES NO WARRANTIES, EXPRESS OR IMPLIED, INCLUDING WITHOUT LIMITATION THE IMPLIED WARRANTIES OF NON-INFRINGEMENT, MERCHANTABILITY AND FITNESS FOR A PARTICULAR PURPOSE, REGARDING THIS TOOL OR ITS USE ALONE OR IN COMBINATION WITH YOUR DEVICES, SYSTEMS, OR SERVICES. BY USING THIS TOOL TO ENABLE TRIM, YOU AGREE THAT, TO THE EXTENT PERMITTED BY APPLICABLE LAW, USE OF THE TOOL IS AT YOUR SOLE RISK AND THAT THE ENTIRE RISK AS TO SATISFACTORY QUALITY, PERFORMANCE, ACCURACY AND EFFORT IS WITH YOU. Are you sure you wish to proceed (y/N)?” • Pretty darned scary, but as long as you have a current backup, and a system like to keep your backups current, you shouldn't worry too much about taking advantage of TRIM to keep your SSD in tip-top shape. • Enter y at the Terminal prompt to enable TRIM, or N to leave TRIM turned off for third-party SSDs. • Once TRIM is enabled, your Mac will need to be rebooted to take advantage of the TRIM service. A Few Additional Notes About TRIM TRIM isn't supported in external enclosures that use or FireWire as the method of connection to your Mac. Enclosures with SSDs do support the use of TRIM. Turning TRIM Off for Third-Party SSDs Should you decide you don’t wish to have TRIM turned on for third-party SSDs, you can use the TRIMforce command to disable TRIM by following the instructions above and replacing the Terminal command with: sudo TRIMforce disable Just as when you turned TRIM on, you'll need to reboot your Mac to complete the process of turning TRIM off. Published: 7/28/2015 Updated. Abstract Murine gammaherpesvirus 68 (MHV68) infection of laboratory strains of mice has provided a tractable small animal model for dissecting gammaherpesvirus pathogenesis. The MHV68 latency associated antigen M2 promotes viral latency establishment in germinal center (GC) B cells and plays an important role in virus infection of plasma cells (PCs), which is linked to virus reactivation. More recently, M2 has been highlighted as a potent immunomodulatory molecule capable of hindering both cell-mediated and humoral immunity to MHV68 infection and subsequent challenges. M2 expression in B cells results in activation of B cell receptor signaling pathways that promote proliferation, differentiation, and cytokine production—a hallmark of gammaherpesviruses. 
In this study, we utilized an adoptive transfer model to explore the biological consequence of M2 expression in activated B cells in vivo. Secondly, we engineered and validated two independent MHV68 M2 reporter viruses that track M2 protein expression in latently infected B cells during infection. Here we demonstrate that upon adoptive transfer into naive mice, M2 expression promotes activated primary B cells to competitively establish residency in the spleen as either a GC B cell or a PC, most notably in the absence of an ongoing GC reaction. Moreover, M2 antigen drives robust PC differentiation and IL10 production in vivo in the absence of other viral factors. Lastly, we confirm that M2 expression during MHV68 infection is localized to the GC compartment, which is a long term latency reservoir for gammaherpesviruses. Overall, these observations are consistent with, and extend upon previous reports of M2 function in B cells and within the context of MHV68 infection. Moreover, this work provides support for a model by which M2-driven dysregulation of B cell function compromises multiple aspects of antiviral immunity to achieve persistence within the infected host. Author summary Gammaherpesvirus (GHVs), which primarily infect B cells, are capable of exploiting B cell biology to achieve a stable and persistent infection for the lifetime of the host. GHV infections traffick to germinal center (GC) B cells and plasma cells (PCs), which are important immune effectors that promote the generation of protective antibodies in response to pathogens. The mechanism by which murine gammaherpesvirus 68 (MHV68) M2 latency protein activates B cell receptor signaling pathways to modulate the immune response to infection and further promote viral pathogenesis within the GC B cell and PC compartments is not completely understood. Here we demonstrate that M2 expression alone, in the absence of other viral factors, drives robust PC differentiation and IL10 production in vivo. Moreover, M2 promotes the accumulation of splenic GC B cells, which was subsequently verified as the site for potent M2 expression during latent MHV68 infection. Our work further substantiates a model in which a viral protein dysregulates B cell activation, differentiation, and cytokine production to create a permissive environment for viral persistence in the infected host. This work justifies further investigations addressing the impact of GHV latency antigen function within the GC reaction and overall host response to infection. Citation: Terrell S, Speck SH (2017) Murine gammaherpesvirus M2 antigen modulates splenic B cell activation and terminal differentiation in vivo. PLoS Pathog 13(8): e1006543. Editor: Erle S. 
B-cell activation bycrosslinking. Mouse 1gM can bind to B-cell sIgM so as to effect its crosslinking andtherebycanprovide a polyclonal B-cell acti-vating signal. B cell activation and the germinal centre response. Download B cell activation and the germinal centre. Activation signals. Once activated B cells may. B-Cell Tolerance in SLE. B-cell activation depends on positive and negative signals transmitted through the B-cell receptor (BCR) and co-receptors as well as. Jan 23, 2004. B lymphocyte activation relies on signals generated upon recognition of antigen by the B cell antigen receptor (BCR) and on additional signals. 5) including BCR engagement in B-lymphocytes (3, 6, 7), and it has been suggested that PKD negatively regulates BCR signaling by phosphorylating Syk and. 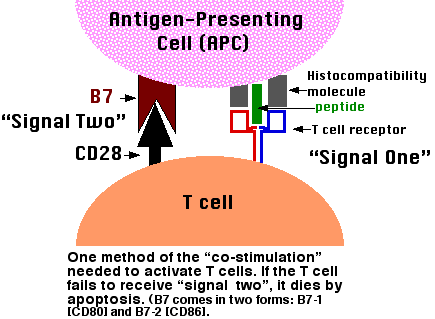 
Robertson, University of Pennsylvania Medical School, UNITED STATES Received: January 30, 2017; Accepted: July 19, 2017; Published: August 2, 2017 Copyright: © 2017 Terrell, Speck. This is an open access article distributed under the terms of the, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Data Availability: All relevant data are within the paper. Funding: This research was funded by the National Institutes of Allergy and Infectious Diseases (grant number R01 AI 058057). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. Competing interests: The authors have declared that no competing interests exist. Validation of M2 reporter constructs that track M2 expression in splenic B cells Extensive characterization of MHV68 M2 antigen function in vitro has revealed a wealth of information, namely that it functions as an adaptor protein by triggering the assembly of multimeric protein complexes that activate various B cell signaling pathways [–, ]. In particular, retroviral transduction of LPS-stimulated primary B cells with a constitutively active M2 expression vector results in enhanced B cell proliferation and IL10 production []. Due to the fact that IL10-dependent cell proliferation requires a functional M2, we utilized this retroviral transduction system as a screening tool to evaluate the detection efficacy of an intracellular marker that could monitor M2 protein expression without adversely disrupting its function. To this end, an M2-mCherry transgene was generated by fusing the M2 ORF upstream of the mCherry fluorescent protein sequence with an intervening 30 amino acid F2A peptide derived from the foot-and-mouth disease virus 2A []. The incorporation of the F2A peptide allows for cotranslation of separate polypeptides from a single mRNA transcript [], thus enhancing the likelihood of preserving both M2 and mCherry functionality. The M2-mCherry construct was cloned into a replication-defective murine stem cell virus (MSCV) vector upstream of an IRES-Thy1.1 cassette, which facilitates detection of transduced B cells by surface Thy1.1 expression () []. Primary B cells were isolated from bulk splenocytes by negative selection and stimulated overnight with LPS prior to transduction with the MSCV-M2-mCherry retrovirus, or the previously described wild type MSCV-M2 and negative control MSCV-M2.stop retroviruses (the latter contains a translation stop codon at amino acid 13 of the M2 open reading frame) (). 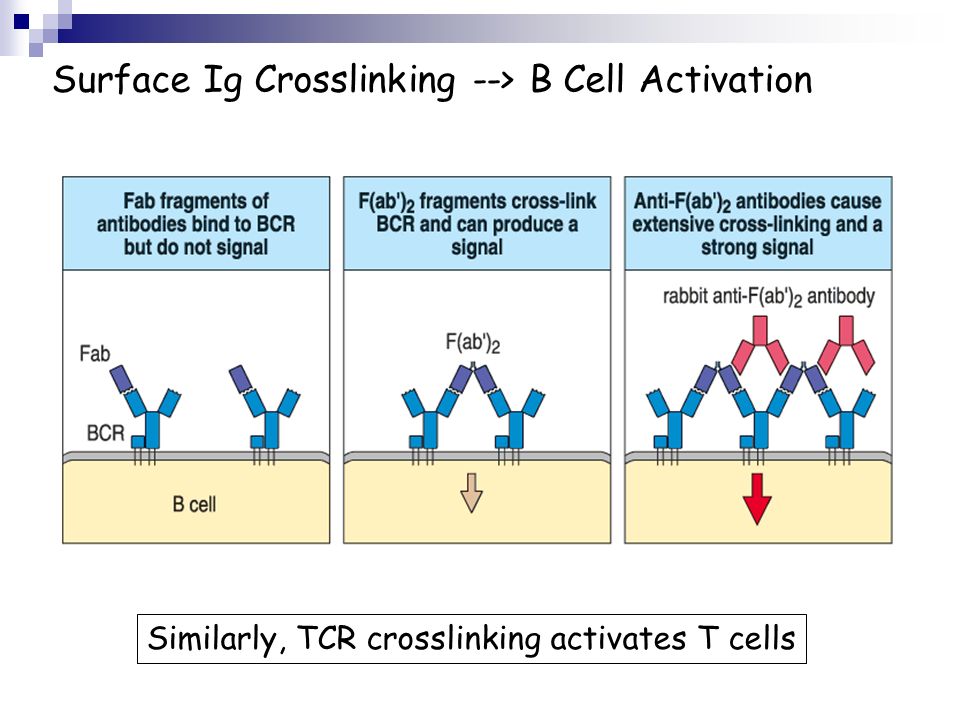
Following retroviral transduction, B cell cultures were harvested in triplicate at each time point and monitored for cell surface Thy1.1 expression and IL10 production. Consistent with previously published reports [, ], transduced cells (Thy1.1+) expressing M2 expanded to ~90% of the culture and secreted ~40ng/mL of IL10 by day five post-transduction, which was not observed in M2.stop-transduced cultures (). M2-mCherry expressing cultures exhibited Thy1.1+ B cell expansion and robust IL10 production nearly identical to that observed with the wild type M2 expression construct (), indicating that M2 protein expressed from the fusion gene possessed wild type M2 function in B cells. Detection of M2 reporter activity in activated splenic B cells. (A) Representative images of transduced primary B cells obtained from brightfield and TRITC filters at day 3 post-transduction. (B) Representative flow cytometry histograms obtained at day 3 post-transduction compare mCherry expression in Thy1.1 positive (red) and Thy1.1 negative (grey) populations as determined by FACS analysis. (C) Frequencies of cell surface Thy1.1 and intracellular mCherry expression of M2-transduced (left panel) and M2-mCherry-transduced (right panel) B cell cultures were monitored over the time course by flow cytometry. Mean and standard deviation values were derived from compiling data from two independent experiments containing three replicates per condition per time point. M2 antigen expression modulates splenic B cell activation and differentiation in vivo. The transduced adoptively transferred B cell populations from M2 and M2.stop animals were analyzed for cell surface marker expression as a function of transgene expression. Data is representative of two independent experiments with 3 mice/group. A) Representative histogram depicting Thy1.1 expression within the adoptively transferred (CD45.1+) population in M2 and M2.stop animals at D5 post transfer. Thy1.1 expression in the CD45.1+ fraction was divided into Thy1.1 –, Thy1.1 Lo/Int (or Thy1.1 +), and Thy1.1 Hi populations. Representative histograms displaying B cell surface marker expression and intracellular IL10 production corresponding to Thy1.1 expression intensities are displayed for M2 (B) and M2.stop (C) transduced B cells. Recombinant MHV68-H2bYFP M2 reporter viruses efficiently establish latency and reactivate infectious virus from splenic B cells Given that the phenotype of adoptively transferred M2-transduced B cells was consistent with MHV68 latency reservoirs in vivo, we sought to interrogate the context of M2 expression during infection. To this end, we generated two independent M2 reporter viruses in the background of the previously described MHV68-H2bYFP virus, which marks latently infected B cells with intracellular YFP that can be detected by flow cytometry []. Previous studies demonstrated that YFP+ splenic B cells contain the viral genome and are capable of reactivating infectious virus, which allows us to utilize YFP expression as an internal control for latency establishment efficiency. To track M2 expression under the control of its native promoter, the M2 ORF in the context of the MHV68-H2bYFP BAC genome was substituted with either the M2-mCherry or the M2-Thy1.1 reporter construct sequences via the galK recombination system () []. The recombinant MHV68-H2bYFP BAC clones were sequenced at the M2 locus to confirm the presence of the inserted constructs and the integrity of the viral genome was evaluated by RFLP analysis. M2 expression during MHV68 infection in vivo enhances latency establishment and virus reactivation in a dose and route dependent manner [, ]. To evaluate overall viral fitness and account for any deleterious genomic alterations, we sought to quantify latency establishment efficiencies for the recombinant MHV68-H2bYFP M2 reporter viruses. C57BL/6 mice were infected at 1000 PFU via the intraperitoneal (IP) route with the parental MHV68-H2bYFP virus and two independent clones of either the M2-mCherry or M2-Thy1.1 reporter viruses and intracellular YFP expression in latently infected splenocytes was detected by flow cytometry. Consistent with previous reports, the H2bYFP virus displayed a typical variation in the frequency of latently infected B cells (%B220 +YFP +) that averaged ~0.3% at 14dpi () [, ]. M2-mCherry- and M2-Thy1.1- infected animals exhibited on average ~0.2% and ~0.3% B220 +YFP + cells, respectively, which was not significantly different to that of the parental MHV68-H2bYFP virus (). We subsequently evaluated virus reactivation from latently infected splenocytes upon explant into tissue culture by utilizing the previously described limiting dilution ex vivo reactivation assay []. The average frequency of cells reactivating infectious virus following M2-mCherry (1 in 6,309 cells) and M2-Thy1.1 (1 in 5,128 cells) infection was similar to that of parental H2bYFP (on average 1 in 6,165 cells) (). In conclusion, our extensive evaluation of the recombinant MHV68-H2bYFP M2 reporter viruses verify that the addition of exogenous sequences at the M2 locus did not dramatically alter M2-driven latency establishment in vivo and subsequent ex vivo virus reactivation from latently infected splenic B cells. Generation of recombinant MHV68 M2 reporter viruses. (A) The M2-Thy1.1 or M2-mCherry reporter construct () was incorporated at the M2 locus in the MHV68 H2bYFP background under the control of the native M2 promoter. C57BL/6 mice were infected at 1000PFU via the intraperitoneal route and splenocytes were harvested at 14 dpi. Statistics were determined by two-tailed unpaired t test with Welch’s correction. (B and C) Representative flow plots (left) and quantitation (right) of B220 +YFP + frequency in latently infected splenocytes were obtained from infections comparing the parental MHV68 H2bYFP virus and two (2) independent clones of either the MHV68 H2bYFP M2-mCherry (B) or M2-Thy1.1 (C) virus. Each data point represents one animal and the horizontal bar represents the mean. M2 reporter viruses efficiently reactivate virus from latently infected splenocytes ex vivo. Mice were infected at 1000PFU IP with the parental MHV68 H2bYFP virus and two independent clones of either the MHV68 H2bYFP M2-mCherry (A) or M2-Thy1.1 (B) viruses. Infected splenocytes harvested at 14 dpi were pooled from 3–5 mice/condition and serial dilutions were plated onto feeder cells as described in materials and methods. Cytopathic effect (CPE) was scored at 14–21 days post-explant to determine the frequency of cells that are capable of reactivation infectious virus ex vivo. M2 reporter activity is localized to GC B cells during latent MHV68 infection Previous studies characterizing the MHV68-H2bYFP virus demonstrated that a majority of latently infected B cells faithfully display either a GC or a PC phenotype []. Incorporation of the M2.stop mutation in the MHV68-H2bYFP background revealed that M2 expression was dispensable for access to the GC B cell compartment, but critical for viral entry into the PC compartment that serves as a major reservoir for reactivating virus []. Accordingly, we sought to analyze the cell surface phenotype of M2-expressing B cells during latency establishment in vivo. To facilitate a side by side comparison of the engineered MHV68-H2bYFP M2 reporter viruses, animals were infected at 1000 PFU IP with either the parental H2bYFP virus, M2-mCherry virus, or M2-Thy1.1 virus. First, we quantified the M2 reporter positive population within the latently infected B220 +YFP + population by utilizing the parental H2bYFP virus as a negative control. Following IP infection, we detected a robust population of cells that was positive for intracellular mCherry or cell surface Thy1.1 expression with the respective reporter virus, which was not present in H2bYFP-infected animals (). Very similar results were obtained with M2-mCherry and M2-Thy1.1 reporter virus infection, which resulted in ~40% of the latently infected B cell population exhibiting detectable M2 reporter expression at 14 dpi (). Latently infected B220 +YFP + B cells characteristically exhibit a GC phenotype during latent MHV68 H2bYFP infection, which was recapitulated in M2 reporter virus infected animals (, upper panels). Accordingly, ≥90% of mCherry+ and Thy1.1+ cells within the B220 +YFP + compartment reproducibly and almost exclusively exhibited a GC phenotype (, lower panels). Latently infected populations were also analyzed for cell surface markers consistent with a PC phenotype, which comprises ~20% of the CD3 -YFP + population (, upper panels). Although both reporter viruses were capable of efficiently establishing latency within the PC reservoir, we were consistently unable to detect M2 reporter activity in latently infected PCs using either the M2-mCherry or M2-Thy1.1 viruses (, lower panels). Robust M2 reporter expression is observed in a fraction of latently infected B cells in vivo. Mice were infected at 1000PFU IP with MHV68 H2bYFP and second generation MHV68 H2bYFP M2 reporter viruses M2-mCherry and M2-Thy1.1. Splenocytes were harvested at 14 dpi and analyzed for cell surface marker and fluorescent protein expression by flow cytometry. Each data point represents one animal and the horizontal bar represents the mean. Statistics were determined by one way analysis of variance (ANOVA) followed by Bonferonni’s multiple comparisons post tests. (A and B) Representative flow plots and quantitation of intracellular mCherry (A) or cell surface Thy1.1 (B) detection within the latently infected B cell compartment (CD3 -B220 +YFP +) of the indicated virus-infected animals. M2 protein is detected in the germinal center but not in the plasma cell compartment during latency. Mice were infected at 1000PFU via the intraperitoneal route with the parental MHV68 H2bYFP and second generation MHV68 H2bYFP M2 reporter viruses. Splenocytes were harvested and analyzed by flow cytometry at 14 dpi. Each data point represents one animal and the horizontal bar represents the mean. Statistics were determined by one way analysis of variance (ANOVA) followed by Bonferonni’s multiple comparisons post tests. (A) Representative flow plots (left) and quantitation (right) of latently infected B220 +YFP + population (top panel) and B220 +YFP +M2-reporter + cells (bottom panel) exhibiting a germinal center phenotype (GL7 HiCD95 Hi). (B) Representative flow plots (left) and quantitation (right) of YFP+ (top panel) and YFP +M2-reporter + cells (bottom panel) exhibiting a plasma cell phenotype (B220 LoCD138 Hi). We subsequently evaluated MHV68 H2bYFP M2-Thy1.1 infection following inoculation at 1000 PFU IN, which represents a more stringent route of infection, in order to reveal any potential defects with viral trafficking to the spleen. Analysis of infected splenocytes at 16 dpi via the IN route demonstrated that the M2-Thy1.1 virus exhibited no significant alterations in latency establishment or viral trafficking to the GC and PC compartments (). M2 reporter positive cells comprised on average ~20% of the B220 +YFP + population (), which was modestly reduced compared to the IP route (~40%; ). Despite a slightly lower frequency of YFP +Thy1.1 + cells, we found that M2 reporter activity was reproducibly detected within the GC, but not the PC compartment (), which is consistent with the results obtained following IP infections (). In conclusion, our M2 reporter viruses system has clearly established the latently infected GC B cell compartment as a critical site of potent M2 expression during latent MHV68 infection in vivo. M2 expression is predominantly associated with a germinal center phenotype following intranasal infection. Mice were infected at 1000 PFU via the intranasal route with the parental MHV68 H2bYFP and MHV68 H2bYFP M2-Thy1.1 viruses. Splenocytes were harvested and cell surface markers were analyzed by flow cytometry at 16 dpi. Each data point represents one animal and the horizontal bar represents the mean. Statistics were determined by two-tailed unpaired t test with Welch’s correction. The frequency of B220 +YFP + latently infected B cells (A) exhibiting a germinal center (B) or plasma cell (C) phenotype were identified by flow cytometry as described in. The B220 +YFP +Thy1.1 + (M2 reporter) population was identified as described in and subphenotyped using surface markers consistent with a germinal center B cell (E) and plasma cell (F). Discussion Prior studies have demonstrated a critical role for MHV68 M2 with respect to viral latency establishment and virus reactivation in a mouse model of infection [–]. In this study, we utilized complementary methods to further elucidate the context and potential impact of M2 expression with respect to B cell function in vivo. Our adoptive transfer studies are the first demonstration that M2 drives robust PC differentiation and IL10 production in vivo in the absence of other viral factors (Figs and ). However, we were unable to detect M2 reporter activity in the latently infected PC compartment (Figs and ), which may indicate that the M2 promoter is no longer active once the cell has reached a sufficient level of IRF4 expression required for terminal differentiation into a PC []. Additionally, the timing and low level of M2 protein expression that is required for PC differentiation, as demonstrated in our adoptive transfer system (), may hinder our detection of M2 protein at 14 dpi in the M2 reporter virus system (Figs and ). Although we were unable to directly confirm protein expression in the latently infected PC, the adoptive transfer data fits into a well-established model in which M2-driven IRF4 expression facilitates PC differentiation during MHV68 infection [,,, ]. A significant portion of latently infected PCs can be generated via the extrafollicular pathway [] and we have shown here that M2 drives robust PC differentiation in the absence of an ongoing GC reaction (Figs and ). How do latently infected PCs contribute to GHV pathogenesis? PC differentiation initiates lytic replication of EBV and KSHV via activation of viral transactivators by PC-specific transcription factors such as XBP-1 and Blimp-1 [,, –]. At least one study has reported that de novo KSHV infection drives human tonsillar B cells to proliferate and differentiate into plasmablasts that functionally and phenotypically resemble multicentric Castleman’s disease []. Therefore, PC differentiation represents a common aspect of GHV pathogenesis and this mechanism of virus reactivation has been proposed to facilitate virus transmission and maintenance of a stable life-long infection. For MHV68, PCs have been identified as the predominant source of infectious virus production, and virus reactivation is severely impaired in the absence of M2 []. Moreover, PCs appear to play an important role in virus trafficking and seeding of chronic MHV68 latency reservoirs. For example, an M2-null virus exhibits significantly impaired latency establishment in the spleen at 16 dpi, despite efficient viral replication in the lungs following low dose intranasal inoculation. Additionally, long term latency maintenance at 90 dpi was severely attenuated in mice lacking splenic PCs in a conditional Blimp-1 knockout model. In combination, our studies support a model in which GHVs play a direct role in driving PC differentiation which serves to facilitate reactivation of infectious virus and reseeding latency reservoirs within the infected host. In addition to directly promoting virus reactivation, IL10 production by PCs has the potential to suppress humoral immunity and create a more permissive environment for viral infection. IL10 is a potent immunomodulatory cytokine that impairs T cell, macrophage and dendritic cell functions in a variety of infection settings (reviewed in []). GHVs exploit the IL10 signaling pathway by encoding viral IL10 homologs and/or enhancing IL10 expression from B cells, which promotes B cell expansion and abrogates immune recognition and subsequent eradication of infected B cells in vitro [, –]. During primary MHV68 infection M2 functions as an immunomodulatory molecule by elevating serum IL10 levels, attenuating antiviral CD8 T cell responses, and suppressing antigen-specific responses to MHV68 and subsequent challenges[, ]. Here we have shown that M2-driven PC differentiation is characterized by robust CD138 expression and IL10 production (), which is consistent with M2-driven IRF4 production in B cells []. Interestingly, B cell IL10 production also attenuates aspects of innate and adaptive immunity in a salmonella infection model, and IRF4 HiCD138 Hi PCs have been identified as a potent source of IL10 [, ]. Therefore, we propose that latently infected PCs could serve as one potential source of immunsuppressive IL10 production during MHV68 infection. Moreover, dysregulated BCR signaling may represent a common mechanism by which GHVs and other pathogens promote regulatory PC generation as an immune evasion tactic during infection. The ultimate significance of IL10 expression in the context of GHV infection in vivo is still under debate, and investigations are currently underway to evaluate the contribution of host-derived IL10 to MHV68 pathogenesis. The prevailing model of GHV pathogenesis requires that the virus traverse the GC compartment in order to gain access to the long-lived memory B cell compartment (reviewed in []). The GC reaction represents a competitive environment wherein B cells that do not receive rescue signals triggered by antigen recognition or T cell help are subjected to death by apoptosis while high affinity B cells that successfully compete for limited T cell help are positively selected to enter the long-lived B cell compartment [, ]. Similar to EBV, MHV68-latently infected B cells resemble, localize and participate in ongoing GC reactions. Importantly, and in contrast to EBV, T cell help is a demonstrated requirement for expansion of latently infected GC B cells and entry to the memory B cell pool, which serves as the long term latency reservoir for GHVs [,,, ]. M2 transcripts have been detected in GC B cells during chronic infection [, ] and our M2 reporter virus system has independently verified that the latently infected GC compartment is a site of robust M2 antigen expression at the peak of MHV68 latency, which was independent of the route of virus inoculation (Figs and ). In an increasingly hostile environment, M2 antigen expression could promote latently infected B cell survival and exit from the GC as a memory B cell. In this study, we show that M2 antigen expression in stimulated B cells was able to support, at least temporarily, the activated GC phenotype in the absence viral infection (Figs and ). Moreover, M2-driven signaling promotes the formation of B-T cell conjugates in the presence and absence of specific peptide [], which in the context of the GC reaction could provide sufficient CD40 stimulation to enhance survival and selection of latently infected B into the memory B cell pool [–]. Further investigation is necessary in order to determine if M2 expression in GC B cells promotes viral trafficking to the memory B cell pool in vivo. Previous reports have confirmed the presence of M2 transcripts in memory B cells [], but these analyses are inherently misleading and more sensitive and quantitative analyses are required to correlate MHV68 transcriptional programs with specific stages of B cell differentiation. Thus, enhanced characterizations of MHV68 latency antigen function and transcriptional programs may reveal common strategies by which GHVs effectively manipulate GC B cell biology to achieve short and/or long term persistence in vivo. In conclusion, our studies have further validated a model in which M2 antigen expression dysregulates B cell activation, differentiation, and cytokine production to simultaneously thwart immune detection and eradication and promote MHV68 pathogenesis in the infected host. While the role of M2 expression within the GC B cell remains unknown, it has great potential to significantly influence both B and T cell responses to MHV68 infection. Therefore, our studies justify continued investigations that address the impact of M2 expression with respect to the global GC response during primary and secondary infections, as this may provide important insights with respect to GHV pathogenesis and associated disease. Virus and tissue culture maintenance BAC-derived MHV68 viruses were reconstituted following transfection of Vero-Cre cells, a generous gift provided by David Lieb []. Recombinant MHV68 viruses were propagated and titered on NIH3T12s (ATCC: CCL-164) as described previously []. Murine embryonic fibroblasts (MEFs) utilized in ex vivo reactivation assays were isolated from day 16 C57BL/6J embryos cultured as previously described []. Adherent cell lines were maintained in Dulbecco’s modification of Eagle medium (DMEM) supplemented with 10% fetal bovine serum, 2mM L-glutamine, and 100U penicillin and 100mg streptomycin per mL. Primary B cells isolated from C57BL/6J mice were maintained in Roswell Park Memorial Institute (RPMI) 1640 Medium supplemented with 10mM non-essential amino acids, 1mM sodium pyruvate and 10mM HEPES. Plasmids and retroviral constructs Plasmid MSCV-M2-IRES-Thy1.1 (MSCV-M2) and MSCV-M2.stop-IRES-Thy1.1 (MSCV-M2.stop) have been previously described []. Flow cytometry Blocking and detection antibodies were diluted in PBS supplemented with 2% FBS and 1mM EDTA. Splenocytes were blocked with anti-CD16/32 (BD bioscience) for 15 minutes on ice prior to surface staining for 30 minutes on ice. Antibodies used in this study: B220-Pac Blue, CD138-BV650 and -APC, CD45.1-FITC, CD3-PerCp, CD4-PerCp, CD8-PerCp, Thy1.1-PE, CD95-PE/Cy7, GL7-APC, and CD19-Pac Blue, -BV650, -FITC,- PerCp, -PE, -PE/Cy7, -APC, -APC/Cy7, -Alexa Fuor 594 (BD bioscience, eBioscience, or Biolegend). For intracellular cytokine staining, unstimulated cells were fixed with 4% paraformaldehyde/PBS solution after surface staining step. Cells were subsequently permeabilized using the BD Cytofix/Cytoperm Fixation/Permeabilization Kit (BD biosciences) per manufacturer’s instructions prior to staining with IL10-PE/Cy7 (Biolegend). Dead cells were labeled with fixable viability dye eFluor780 (eBioscience) per manufacturer’s instructions. Cells were analyzed on a BD LSRII flow cytometer and data was analyzed using FlowJo software. Construction of recombinant viruses M2-mCherry H2bYFP and M2-Thy1.1 H2bYFP bacterial artificial clones (BACs) were generated utilizing the galK selection method []. The M2 locus in the background of the previously described recombinant MHV68-H2bYFP genome [] was replaced with galK gene as previously described []. Briefly, the galK cassette was amplified with primers flanked with 50bp sequence homology to the target sequence (5’-aggcgtgtttaaagaaaaagttatgttctgcgtta gcaccttcactgttacctgttgacaattaatcatcggca-3’ and 5’-agggggtttcaacaggcactagtctgatgaggtttcgtttt caggtaatgtcagcactgtcctgctcctt-3’) prior to electroporation of SW102 cells harboring the MHV68 H2bYFP BAC. The M2/galK intermediates exhibiting galactokinase activity were selected as previously described [] and the presence of the desired insertion at the M2 locus was confirmed by restriction fragment length polymorphism (RFLP) analysis. M2-mCherry and M2-Thy1.1 sequences were amplified from the MSCV-M2-mCherry and MSCV-M2 vectors, respectively, with primers containing the 50bp homology arms and each cassette was electroporated into SW102 cells harboring the M2/galK intermediate. Positive recombinants were identified by PCR colony screen for the presence of the desired sequence and further evaluated by sequencing and RFLP analysis. Statistical analysis GraphPad Prism software (San Diego, CA) was used to generate data graphs and perform statistical analyses. For line and bar graphs, the mean and standard deviation were plotted for each condition in triplicate. For scatter plots, each data point represents on animal and the horizontal bar represents the mean. Statistical significance between two conditions was determined by two-tailed unpaired t test with Welch’s correction. For three or more conditions, statistical significance was evaluated by one way analysis of variance (one way ANOVA) analysis followed by Bonferonni’s multiple comparisons post-tests. For reporting of absolute numbers of B cell populations in adoptive transfer recipients, 1 cell was added to all values in order to avoid the undefined logarithm of zero. References • 1. McGeoch DJ, Rixon FJ, Davison AJ. Topics in herpesvirus genomics and evolution. Pmid:16490275 • • • • 2. Chang Y, Cesarman E, Pessin MS, Lee F, Culpepper J, Knowles DM, et al. Identification of Herpesvirus-Like DNA-Sequences in Aids-Associated Kaposis-Sarcoma. Pmid:7997879 • • • • 3. Henle G, Henle W, Clifford P, Diehl V, Kafuko GW, Kirya BG, et al. Antibodies to Epstein-Barr Virus in Burkitts Lymphoma and Control Groups. J Natl Cancer I. Virgin HW, Latreille P, Wamsley P, Hallsworth K, Weck KE, DalCanto AJ, et al. Complete sequence and genomic analysis of murine gammaherpesvirus 68. Journal of Virology. Pmid:9223479 • • • • 5. Flano E, Kim IJ, Woodland DL, Blackman MA. Gamma-herpesvirus latency is preferentially maintained in splenic germinal center and memory B cells. PubMed Central PMCID: PMCPMC2193987. Pmid:12438427 • • • • 6. Sunilchandra NP, Efstathiou S, Nash AA. Murine Gammaherpesvirus 68 Establishes a Latent Infection in Mouse B Lymphocytes Invivo. Pmid:1469366 • • • • 7. Willer DO, Speck SH. Long-term latent murine gammaherpesvirus 68 infection is preferentially found within the surface immunoglobulin D-negative subset of splenic B cells in vivo. Journal of Virology. Pmid:12857900 • • • • 8. Karupiah G, Sacks TE, Klinman DM, Fredrickson TN, Hartley JW, Chen JH, et al. Murine cytomegalovirus infection-induced polyclonal B cell activation is independent of CD4(+) T cells and CD40. Pmid:9448685 • • • • 9. Stevenson PG, Doherty PC. Non-antigen-specific B-cell activation following murine gammaherpesvirus infection is CD4 independent in vitro but CD4 dependent in vivo. Journal of Virology. Pmid:9882308 • • • • 10. Thorleylawson DA, Mann KP. Early Events in Epstein-Barr Virus-Infection Provide a Model for B-Cell Activation. Journal of Experimental Medicine. Pmid:2989413 • • • • 11. Caldwell RG, Wilson JB, Anderson SJ, Longnecker R. Epstein-Barr virus LMP2A drives B cell development and survival in the absence of normal B cell receptor signals. Uchida J, Yasui T, Takaoka-Shichijo Y, Muraoka M, Kulwichit W, Raab-Traub N, et al. Mimicry of CD40 signals by Epstein-Barr virus LMP1 in B lymphocyte responses. Swanson-Mungerson M, Bultema R, Longnecker R. Epstein-Barr virus LMP2A enhances B-cell responses in vivo and in vitro. Pmid:16809282; PubMed Central PMCID: PMCPMC1489056. Babcock GJ, Hochberg D, Thorley-Lawson AD. The expression pattern of Epstein-Barr virus latent genes in vivo is dependent upon the differentiation stage of the infected B cell. Roughan JE, Thorley-Lawson DA. The intersection of Epstein-Barr virus with the germinal center. Pmid:19193789; PubMed Central PMCID: PMCPMC2663245. Pires de Miranda M, Lopes FB, McVey CE, Bustelo XR, Simas JP. Role of Src homology domain binding in signaling complexes assembled by the murid gamma-herpesvirus M2 protein. Pmid:23258536; PubMed Central PMCID: PMCPMC3567640. Rangaswamy US, Speck SH. Murine gammaherpesvirus M2 protein induction of IRF4 via the NFAT pathway leads to IL-10 expression in B cells. Pmid:24391506; PubMed Central PMCID: PMCPMC3879372. De Miranda MP, Alenquer M, Marques S, Rodrigues L, Lopes F, Bustelo XR, et al. The Gammaherpesvirus m2 Protein Manipulates the Fyn/Vav Pathway through a Multidocking Mechanism of Assembly. ARTN e1654 pmid:18301737 • • • • 19. Siegel AM, Herskowitz JH, Speck SH. The MHV68 M2 protein drives IL-10 dependent B cell proliferation and differentiation. Pmid:18389062; PubMed Central PMCID: PMCPMC2270344. Herskowitz JH, Jacoby MA, Speck SH. The murine gammaherpesvirus 68 M2 gene is required for efficient reactivation from latently infected B cells. Pmid:15681428; PubMed Central PMCID: PMCPMC546582. Jacoby MA, Virgin HWt, Speck SH. Disruption of the M2 gene of murine gammaherpesvirus 68 alters splenic latency following intranasal, but not intraperitoneal, inoculation. PubMed Central PMCID: PMCPMC135904. Pmid:11799175 • • • • 22. Liang X, Collins CM, Mendel JB, Iwakoshi NN, Speck SH. Gammaherpesvirus-driven plasma cell differentiation regulates virus reactivation from latently infected B lymphocytes. Pmid:19956661; PubMed Central PMCID: PMCPMC2777334. Laichalk LL, Thorley-Lawson DA. Terminal differentiation into plasma cells initiates the replicative cycle of Epstein-Barr virus in vivo. Pmid:15613356; PubMed Central PMCID: PMCPMC538585. Wilson SJ, Tsao EH, Webb BL, Ye H, Dalton-Griffin L, Tsantoulas C, et al. X box binding protein XBP-1s transactivates the Kaposi's sarcoma-associated herpesvirus (KSHV) ORF50 promoter, linking plasma cell differentiation to KSHV reactivation from latency. Pmid:17928342; PubMed Central PMCID: PMCPMC2168861. Fillatreau S, Sweenie CH, McGeachy MJ, Gray D, Anderton SM. B cells regulate autoimmunity by provision of IL-10. Lampropoulou V, Hoehlig K, Roch T, Neves P, Calderon Gomez E, Sweenie CH, et al. TLR-activated B cells suppress T cell-mediated autoimmunity. Mauri C, Gray D, Mushtaq N, Londei M. Prevention of arthritis by interleukin 10-producing B cells. PubMed Central PMCID: PMCPMC2193864. Pmid:12591906 • • • • 28. Neves P, Lampropoulou V, Calderon-Gomez E, Roch T, Stervbo U, Shen P, et al. Signaling via the MyD88 adaptor protein in B cells suppresses protective immunity during Salmonella typhimurium infection. Shen P, Roch T, Lampropoulou V, O'Connor RA, Stervbo U, Hilgenberg E, et al. IL-35-producing B cells are critical regulators of immunity during autoimmune and infectious diseases. Pmid:24572363; PubMed Central PMCID: PMCPMC4260166. Yanaba K, Bouaziz JD, Haas KM, Poe JC, Fujimoto M, Tedder TF. A regulatory B cell subset with a unique CD1dhiCD5+ phenotype controls T cell-dependent inflammatory responses. Lampropoulou V, Calderon-Gomez E, Roch T, Neves P, Shen P, Stervbo U, et al. Suppressive functions of activated B cells in autoimmune diseases reveal the dual roles of Toll-like receptors in immunity. Matsumoto M, Fujii Y, Baba A, Hikida M, Kurosaki T, Baba Y. The calcium sensors STIM1 and STIM2 control B cell regulatory function through interleukin-10 production. Matar CG, Anthony NR, O'Flaherty BM, Jacobs NT, Priyamvada L, Engwerda CR, et al. Gammaherpesvirus Co-infection with Malaria Suppresses Anti-parasitic Humoral Immunity. Pmid:25996913; PubMed Central PMCID: PMCPMC4440701. Marques S, Efstathiou S, Smith KG, Haury M, Simas JP. Selective gene expression of latent murine gammaherpesvirus 68 in B lymphocytes. PubMed Central PMCID: PMCPMC164786. Pmid:12805429 • • • • 35. Virgin HWt, Presti RM, Li XY, Liu C, Speck SH. Three distinct regions of the murine gammaherpesvirus 68 genome are transcriptionally active in latently infected mice. Pmid:9971815; PubMed Central PMCID: PMCPMC104477. Rodrigues L, de Miranda MP, Caloca MJ, Bustelo XR, Simas JP. Activation of vav by the gammaherpesvirus M2 protein contributes to the establishment of viral latency in B lymphocytes. Journal of Virology. Pmid:16731951 • • • • 37. Minskaia E, Nicholson J, Ryan MD. Optimisation of the foot-and-mouth disease virus 2A co-expression system for biomedical applications. Bmc Biotechnol. Artn 67 pmid:23968294 • • • • 38. Collins CM, Boss JM, Speck SH. Identification of infected B-cell populations by using a recombinant murine gammaherpesvirus 68 expressing a fluorescent protein. Pmid:19386718; PubMed Central PMCID: PMCPMC2698576. Warming S, Costantino N, Court DL, Jenkins NA, Copeland NG. Simple and highly efficient BAC recombineering using galK selection. Nucleic Acids Res. Pmid:15731329; PubMed Central PMCID: PMCPMC549575. Collins CM, Speck SH. Tracking murine gammaherpesvirus 68 infection of germinal center B cells in vivo. Pmid:22427999; PubMed Central PMCID: PMCPMC3302828. Rangaswamy US, O'Flaherty BM, Speck SH. Tyrosine 129 of the murine gammaherpesvirus M2 protein is critical for M2 function in vivo. Pmid:25122496; PubMed Central PMCID: PMCPMC4133380. Weck KE, Barkon ML, Yoo LI, Speck SH, Virgin HI. Mature B cells are required for acute splenic infection, but not for establishment of latency, by murine gammaherpesvirus 68. Pmid:8794315; PubMed Central PMCID: PMCPMC190721. Sciammas R, Shaffer AL, Schatz JH, Zhao H, Staudt LM, Singh H. Graded expression of interferon regulatory factor-4 coordinates isotype switching with plasma cell differentiation. Matar CG, Rangaswamy US, Wakeman BS, Iwakoshi N, Speck SH. Murine gammaherpesvirus 68 reactivation from B cells requires IRF4 but not XBP-1. Pmid:25078688; PubMed Central PMCID: PMCPMC4178818. Collins CM, Speck SH. Expansion of murine gammaherpesvirus latently infected B cells requires T follicular help. Pmid:24789087; PubMed Central PMCID: PMCPMC4006913. Reusch JA, Nawandar DM, Wright KL, Kenney SC, Mertz JE. Cellular differentiation regulator BLIMP1 induces Epstein-Barr virus lytic reactivation in epithelial and B cells by activating transcription from both the R and Z promoters. Pmid:25410866; PubMed Central PMCID: PMCPMC4300755. Sun CC, Thorley-Lawson DA. Plasma cell-specific transcription factor XBP-1s binds to and transactivates the Epstein-Barr virus BZLF1 promoter. Pmid:17898050; PubMed Central PMCID: PMCPMC2168822. Yu F, Feng J, Harada JN, Chanda SK, Kenney SC, Sun R. B cell terminal differentiation factor XBP-1 induces reactivation of Kaposi's sarcoma-associated herpesvirus. Hassman LM, Ellison TJ, Kedes DH. KSHV infects a subset of human tonsillar B cells, driving proliferation and plasmablast differentiation. J Clin Invest. Pmid:21245574; PubMed Central PMCID: PMCPMC3026728. Couper KN, Blount DG, Riley EM. IL-10: the master regulator of immunity to infection. Incrocci R, McCormack M, Swanson-Mungerson M. Epstein-Barr virus LMP2A increases IL-10 production in mitogen-stimulated primary B-cells and B-cell lymphomas. 2013;94(Pt 5):1127–33. Pmid:23303827; PubMed Central PMCID: PMCPMC3998239. Jochum S, Moosmann A, Lang S, Hammerschmidt W, Zeidler R. The EBV immunoevasins vIL-10 and BNLF2a protect newly infected B cells from immune recognition and elimination. Pmid:22615564; PubMed Central PMCID: PMCPMC3355093. Moore KW, Vieira P, Fiorentino DF, Trounstine ML, Khan TA, Mosmann TR. Homology of cytokine synthesis inhibitory factor (IL-10) to the Epstein-Barr virus gene BCRFI. Vockerodt M, Haier B, Buttgereit P, Tesch H, Kube D. The Epstein-Barr virus latent membrane protein 1 induces interleukin-10 in Burkitt's lymphoma cells but not in Hodgkin's cells involving the p38/SAPK2 pathway. Thorley-Lawson DA, Hawkins JB, Tracy SI, Shapiro M. The pathogenesis of Epstein-Barr virus persistent infection. Curr Opin Virol. Pmid:23683686; PubMed Central PMCID: PMCPMC3789532. Shulman Z, Gitlin AD, Weinstein JS, Lainez B, Esplugues E, Flavell RA, et al. Dynamic signaling by T follicular helper cells during germinal center B cell selection. Pmid:25170154; PubMed Central PMCID: PMCPMC4519234. Victora GD, Schwickert TA, Fooksman DR, Kamphorst AO, Meyer-Hermann M, Dustin ML, et al. Germinal center dynamics revealed by multiphoton microscopy with a photoactivatable fluorescent reporter. Pmid:21074050; PubMed Central PMCID: PMCPMC3035939. Babcock GJ, Decker LL, Volk M, Thorley-Lawson DA. EBV persistence in memory B cells in vivo. Roughan JE, Torgbor C, Thorley-Lawson DA. Germinal center B cells latently infected with Epstein-Barr virus proliferate extensively but do not increase in number. Pmid:19889783; PubMed Central PMCID: PMCPMC2798379. Fontinha D, Lopes FB, Marques S, Alenquer M, Simas JP. Murid Gammaherpesvirus Latency-Associated Protein M2 Promotes the Formation of Conjugates between Transformed B Lymphoma Cells and T Helper Cells. Pmid:26544979; PubMed Central PMCID: PMCPMC4636232. Klaus GG, Choi MS, Lam EW, Johnson-Leger C, Cliff J. CD40: a pivotal receptor in the determination of life/death decisions in B lymphocytes. Int Rev Immunol. Siepmann K, Skok J, van Essen D, Harnett M, Gray D. Rewiring of CD40 is necessary for delivery of rescue signals to B cells in germinal centres and subsequent entry into the memory pool. PubMed Central PMCID: PMCPMC1783186. Pmid:11298824 • • • • 63. Kim IJ, Flano E, Woodland DL, Lund FE, Randall TD, Blackman MA. Maintenance of long term gamma-herpesvirus B cell latency is dependent on CD40-mediated development of memory B cells. Gierasch WW, Zimmerman DL, Ward SL, Vanheyningen TK, Romine JD, Leib DA. Construction and characterization of bacterial artificial chromosomes containing HSV-1 strains 17 and KOS. J Virol Methods. Clambey ET, Virgin HWt, Speck SH. Disruption of the murine gammaherpesvirus 68 M1 open reading frame leads to enhanced reactivation from latency. Pmid:10644370; PubMed Central PMCID: PMCPMC111675. Lee J, Lee HJ, Shin MK, Ryu WS. Versatile PCR-mediated insertion or deletion mutagenesis. Weck KE, Kim SS, Virgin HI, Speck SH. B cells regulate murine gammaherpesvirus 68 latency. Pmid:10233924; PubMed Central PMCID: PMCPMC112506. Slideshare.net T cells and b- cells 728 × 560 - 182k - jpg slideshare.net T CELL ACTIVATION AND IT'S TERMINATION 638 × 479 - 69k - jpg slideplayer.com TODAY B CELL DEVELOPMENT. - ppt video online download 960 × 720 - 136k - jpg biosiva.50webs.org ANTIBODY PRODUCING CELL 577 × 758 - 91k - jpg slideplayer.com B Cell Generation, Activation, And Differentiation W. 960 × 720 - 133k - jpg pharmrev.aspetjournals. Norepinephrine and β2-Adrenergic Receptor Stimulation Regulate. 781 × 1280 - 165k - jpg embor.embopress.org Activation of B cells by non‐canonical helper signals| EMBO Reports 750 × 649 - 113k - jpg oncohemakey.com B Cell Activation and Signaling| Oncohema Key 509 × 503 - 40k - jpg slideplayer.com B- Cell Generation, Activation, and Differentiation. 960 × 720 - 94k - jpg slideplayer.com Chapter 15 B cell mediated immune response - ppt download 960 × 720 - 70k - jpg medicine.usask.ca Peter Bretscher - College of Medicine - University of Saskatchewan 986 × 533 - 64k - jpg youtube.com B & T Cell Activation & Development, Cytokines, MHC 1 CD40 TNF B7. 1280 × 720 - 74k - jpg slideplayer.com B Cell Activation and the Humoral Immune Response - ppt download 960 × 720 - 101k - jpg what-when-how.com Cell- Cell Interactions, Cytokines, and Chemokines in Immune. 1050 × 2858 - 528k - jpg slideplayer.com Lymphocyte Activation & Immune Tolerance - ppt video online download 960 × 720 - 148k - jpg slideplayer.com C4-11M B Cell Activation and Function III Immunity Mediated by B. 960 × 720 - 78k - jpg slideplayer.com Chapter 11 B- cell Generation, Activation, and Differentiation. 960 × 720 - 112k - jpg slideplayer.com B Cell Tolerance Wendy Davidson Ph.D. May 3, ppt video online download 960 × 720 - 72k - jpg intechopen.com Cells, Molecules and Mechanisms Involved in the Neuro-Immune. 311 × 300 - 15k - jpg slideplayer.com MOLECULAR BASIS OF ANTIGEN RECOGNITION BY B CELLS AND ANTIBODIES. 960 × 720 - 80k - jpg. Abstract • 1ATP can elicit pain in humans and, together with other P2X channel agonists, can produce nocifensive responses in rodents. We used the rat in vitro skin-nerve preparation to quantify primary afferent responses to ATP and its stable analogue,β-methylene ATP in normal and carrageenan-inflamed skin. • 2Both ATP and,β-methylene ATP were found to specifically activate the peripheral terminals of Aδ and C-fibre nociceptors in the skin. Thirty-nine per cent of the nociceptors tested responded to the maximal dose of,β-methylene ATP (5 m m). In contrast, non-nociceptive, low-threshold mechano-sensitive fibres were never activated by the same agonist concentrations. • 3Amongst the nociceptor population, C-mechanoheat fibres (C-MH or polymodal nociceptors) were markedly more responsive to P2X agonists than mechanonociceptors (C-M nociceptors) with Aδ- or C-fibre axons. Both C-mechanoheat and C-mechanonociceptors were activated by,β-methylene ATP doses as low as 50 μ m. • 4In skin inflamed with carrageenan 3-4 h before recording both the number of responsive C-fibre nociceptors and their response magnitude increased. The increased neural response under inflammatory conditions was largely observed in C-mechanoheat or polymodal nociceptors. After low doses of P2X agonists C-MH fibres but not C-M fibres developed elevated ongoing activity and this effect was only seen after carrageenan inflammation. The time course of,β-methylene ATP-evoked discharges in nociceptors was found to correlate well with the time course of behavioural nocifensive responses in rats to the same agonist described in a previous study (). • 5We conclude that the rapid increase in the number of,β-methylene ATP responsive nociceptors and the increased magnitude of the neural response following carrageenan inflammation explains why very low concentrations of such agonists can cause pain in inflammatory states. • • ATP iontophoresed into the skin generates a moderate but dose-dependent burning pain in humans (). In addition, when injected into the hindpaw of rats, ATP also elicits a dose-dependent nocifensive response (Bland-Ward & Humphrey 1997; ). In both rodents and man the threshold concentration for ATP-induced nocifensive responses decreases markedly (up to 100-fold) with a pre-existing inflammatory condition, e.g. 
Carrageenan-inflamed skin in rodents () or blistered skin in humans (). The concentration of extracellular ATP present in some inflamed tissues is also raised ().  
Thus endogenous ATP may reach levels capable of producing pain under pathophysiological conditions (). The P2X ion channels, for which ATP is a ligand, are well suited to mediate the activation of sensory neurons by ATP. Six of the seven members of the P2X ion channel family are present in sensory ganglia () and one member, P2X 3, is selectively expressed by small diameter sensory neurons with unmyelinated axons - presumptive nociceptors (;; ). Patch clamp studies on isolated sensory neurons indicate that ATP elicits inward currents in 40 () to 95% () of the cells, indicating that sensory neurons express functional receptors for ATP (see also Coo). In addition some studies show that isolated sensory neurons identified as nociceptors display distinctive ATP-gated inward currents (Coo; ). Therefore, there is much evidence to support the hypothesis that ATP may activate peripheral nociceptors and produce pain, especially under pathophysiological conditions. Although patch clamp studies are informative about the kinetics of somal ion channel activation, it is of additional interest to determine how the receptive terminals of functionally identified sensory neurons respond to ATP. It is only by recording from individual fibres with functionally characterized terminals that one can judge which subsets of sensory neurons are capable of initiating an ATP nocifensive response. In the present study we have used the in vitro skin-nerve preparation from the rat to characterise the responses of a wide variety of low-threshold mechanoreceptors, mechanonociceptors and polymodal nociceptors to ATP or,β-methylene ATP. The latter agonist is more resistant to tissue hydrolysis and potently activates the P2X 3 and P2X 1 subunits of purinergic ion channels (;;; ). In this study we show that,β-methylene ATP and ATP are capable of evoking sustained discharges specifically from subsets of nociceptors and as such these compounds clearly qualify as algogens. In contrast to studies carried out with isolated neurons, ATP and,β-methylene ATP evoked discharges from single nociceptors that showed little sign of desensitisation during agonist application. We also demonstrate quantitative differences in the sensitivity of different classes of nociceptors to ATP and its analogue. Moreover, these differences persist and are even exacerbated under circumstances where an acute carrageenan inflammation dramatically increases the sensitivity of nociceptor terminals to ATP. METHODS Skin-nerve preparation Adult Wistar rats of either sex were used. Animals were killed by exposure to a rising concentration of CO 2 gas, a method in accordance with German national guidelines. The hair over the hindlimb was shaved and the skin from the area innervated by the saphenous nerve was removed with the nerve intact. After dissection the preparation was placed in an organ bath with the corium side of the skin facing up to ensure efficient oxygenation. The preparation was superfused with an oxygen-saturated modified interstitial fluid solution containing (m m): 123 NaCl, 3.5 KCl, 0.7 MgSO 4, 1.7 NaH 2PO 4, 2.0 CaCl 2, 9.5 sodium gluconate, 5.5 glucose, 7.5 sucrose and 10 Hepes, adjusted to pH 7.4 ± 0.05, temperature, 32 ± 0.5 °C. Recording technique The saphenous nerve was desheathed and individual filaments teased away enabling extracellular recordings to be made from functionally identified single fibres (; ). All data described in this paper are from functionally single fibres. To ensure this was the case a template of the spike under study was saved on the oscilloscope (Tektronix TDS 200) and evoked spikes visually monitored to make sure that no other active spikes would be mistaken for the one under study. All data were collected and saved to disk using Chart software for the Powerlab system running on a PC (ADInstruments). For each single unit the data were analysed off-line using the spike histogram extension of Chart software. This software allows calculation of histograms of spikes discriminated on the basis of a constant height and width. The receptive fields of identified fibres were found by probing the skin with a glass rod. In this way up to 90% of thin myelinated or unmyelinated nociceptors and virtually all of the low-threshold mechanoreceptors can be activated (). Once the borders of the receptive field were determined, a Teflon-coated steel electrode was inserted into the receptive field and the conduction velocity of the afferent determined by electrical stimulation. Units with conduction velocities of 45 °C very shortly after application of the heated solution. Peripheral mediators can contribute to the development and maintenance of inflammatory and neuropathic pain and its concomitants (hyperalgesia and allodynia) via two mechanisms. Activation or excitation by these substances of nociceptive nerve endings or fibers implicates generation of action. View ScienceDirect over a secure connection: switch to HTTPS. Activation of C-nociceptors with capsaicin and neuronal depolarization with KCl also increased. 
Units that responded with more than three spikes were classified as noxious heat-sensitive fibres. On this basis heat-sensitive C-mechanoheat (C-MH) nociceptors were differentiated from heat-insensitive C-mechano- (C-M) nociceptors. Experimental protocol Two experimental protocols were carried out in order to examine different aspects of the response of primary afferents to ATP analogues. (i) High doses of free ATP (10 m m) and,β-methylene ATP (5 m m) were applied to the receptive fields in order to establish the proportion of ATP-sensitive units. The magnitude and duration of responses to these maximal stimuli were also measured. (ii) Increasing doses of,β-methylene ATP (50 μ m-5 m m) were applied to units in order to determine the dose-response relation. The compound was applied for 2 min with a recovery period of 2 min between tests. Carrageenan model of inflammation Animals were deeply anaesthetised with urethane (1.5 g kg −1 i.p.) and four injections of 2% w/v carrageenan (total volume 400 μl) were made intradermally into the area of skin innervated by the saphenous nerve. After 3-4 h the animal was killed by exposure to a rising concentration of CO 2 gas and the skin-nerve preparation was made as described above. In all cases we observed pronounced inflammation and oedema throughout the innervation area of the saphenous nerve after carrageenan injections. Data and statistical analyses A unit was scored as having a response to the agonist even if just one spike more was seen during drug application compared with the preceding control period. This was a realistic criterion as units with very poor responses to low doses most often increased their discharge frequency with subsequently applied increasing doses of agonist. Moreover, many units never responded to any of the agonist concentrations applied. Some nociceptors studied in vitro have low levels of spontaneous activity, and this is increased in inflamed skin (see Results). In order to assess the response to ATP analogues in these units, the baseline level of spontaneous activity (determined in a 2 min pretest period) was subtracted from the response. The response over and above the baseline spontaneous activity was used in all further analyses. The level of spontaneous activity was generally low, and was observed in relatively few fibres. Parametric and non-parametric statistical tests were employed as appropriate. All values are given as means ± s.e.m. A level of 5% was taken as evidence of statistical significance. Compounds used Adenosine 5′-triphosphate lithium salt, and,β-methylene ATP lithium salt were obtained from Sigma. All compounds were dissolved and diluted to the relevant concentration in oxygenated Ringer solution. The pH of each aliquot was adjusted if necessary with sodium hydroxide (40% w/v) to a final value of 7.4. Aliquots were stored at -70 °C and thawed on the day of use. Carrageenan (type IV, Sigma) was dissolved in saline at 2% w/v and was prepared the day before use. RESULTS We recorded from 180 functionally identified sensory fibres in the saphenous nerve of 28 rats. In seven rats the skin innervated by the saphenous nerve was inflamed for 3-4 h prior to the electrophysiological experiments. All the major types of sensory receptors were sampled but the emphasis in this study was on nociceptors. The sample included 126 C-fibres (conduction velocity. • • Examples of responses to,β-methylene ATP A, response of a single C-MH fibre to,β-methylene ATP (5 m m, 5 min). B, example of an Aδ-nociceptor responding to a 2 min application of,β-methylene ATP (5 m m) (left panel). None of the Aβ-fibres tested responded to application of,β-methylene ATP (5 m m); an example is shown in the right panel. At the end of the wash-out period the A-fibre was tested with a mechanical stimulus (*) to confirm that the spike shape was unchanged. In C the average C-MH fibre response (n = 12 different fibres) to a 5 min application of,β-methylene ATP (5 m m) is depicted as a spike frequency histogram. Note that the mean response desensitises in the continued presence of the agonist. The mean magnitude of the response elicited in different subtypes of nociceptors was also quantified. C-MH fibres exhibited the largest mean discharge in response to,β-methylene ATP, averaging 95.9 ± 19.1 spikes over a 2 min test period. This was almost double the average Aδ-nociceptor response at 49.7 ± 27.7 spikes (2 min) −1 and significantly greater than the average C-M response of 25.3 ± 11.8 spikes (2 min) −1 (Mann-Whitney U test, P 0.3). Only eight Aδ-fibre nociceptors were tested at all doses of,β-methylene ATP and therefore the number of responding units (n = 2) was too small to make a meaningful dose-response analysis. • • Dose-response relationships of C-fibre nociceptors to,β-methylene ATP A and B show the responses of C-MH fibres. In A the dose-related increase in the magnitude of the response of C-MH fibres in normal (^) and inflammatory (□) conditions is shown (two-way ANOVA, P. • • C-fibres exhibit tachyphylaxis to,β-methylene ATP The time course of the response to the same chemical stimulus of a naive population of C-MH units (□) and the response of a population of C-MH units previously exposed to lower doses of,β-methylene ATP (50 μ m and 0.5 m m, ^) are shown (n = 10-12). Note that after previous exposure to the agonist the mean response magnitude is reduced throughout the agonist application. Effects of acute carrageenan inflammation We recorded from 54 afferents innervating the inflamed skin from seven animals. There was an increase in the spontaneous activity of units innervating carrageenan-inflamed skin, as reported in previous studies (; ). In the absence of prior chemical stimulation the mean resting discharge of both C-MH and C-M fibres was higher than in controls (0.79 ± 0.50 compared with 0.13 ± 0.04 spikes s −1). The level of spontaneous activity was calculated and subtracted from the discharge elicited during the application of the agonist in order to specifically assess the response to ATP analogues (see Methods). A greater proportion of C-fibres innervating inflamed skin responded to,β-methylene ATP compared with the proportion of responsive C-fibres in normal skin. Thus, 43% of C-fibres (12/30 units) innervating normal skin responded to 50 μ m,β-methylene ATP compared with 70% of C-fibres (31/44 units) innervating inflamed skin (χ 2 test, P. • • Dose-dependent activation of C-MH and C-M fibres by,β-methylene ATP in normal and inflamed skin In A the time course of C-MH fibre activation by,β-methylene ATP (n = 16-21) is shown; spontaneous activity was not subtracted in this case. For clarity the activity between agonist doses is not shown (note the breaks in the abscissa). Note that in inflamed skin (□) low doses of,β-methylene ATP induce a long-lasting increase in on-going activity in C-MH fibres that was not observed in normal skin (^). Thus the mean activity is already high in these fibres shortly before application of the second and third doses of,β-methylene ATP. In B the equivalent data from C-M fibres are plotted (n = 5-15). Note that these fibres are much less affected by carrageenan inflammation and no significant effect was observed of low doses of agonist on on-going activity. DISCUSSION Using the rat in vitro skin-nerve preparation we show here that agonists of P2X receptors selectively activate the peripheral terminals of nociceptive sensory neurons in the skin. Primary afferent neurons with non-nociceptive properties, e.g. Low-threshold mechanoreceptors, were never activated by either high concentrations of free ATP or,β-methylene ATP. We could also show that specific types of nociceptors respond quite differently, both qualitatively and quantitatively, to P2X agonists. Specifically noxious heat sensitive C-fibre nociceptors (or polymodal nociceptors) responded more robustly in a dose-dependent manner to,β-methylene ATP, and this response was greatly potentiated by a pre-existing carrageenan inflammation. In contrast, C-fibre mechanonociceptors and A-fibre nociceptors responded poorly to P2X agonists and prior inflammation of the skin did not increase the response of individual C-M fibres to lower doses of,β-methylene ATP (). However, at the highest dose of,β-methylene ATP used (5 m m) the response of C-M fibres was increased in inflamed skin, but in absolute terms the increase in mean discharge of C-M fibres in inflamed skin never approached that of C-MH fibres ( and ). It is possible that the apparently selective effects that we see of P2X agonists in activating the receptive terminals of certain types of nociceptors are in fact indirect. Thus some of the effects could be due to the activation of other cell types (mast cells, blood vessels or sympathetic terminals) that in turn release substances that excite nociceptors. However, the expression of functional P2X-gated ion channels in sensory neurons most probably means that the agonists we have used exert their effects directly. Further experiments would be required to include or exclude indirect activation by other skin cell types. The demonstration here that P2X agonists selectively excite physiologically defined nociceptors explains why ATP elicits pain in humans when applied to the periphery (;; ). Indeed the time course and magnitude of nociceptor activation correlate remarkably well with nocifensive responses in rats and pain scores in humans following peripheral application of P2X agonists (). In a recent study Kress & Guenther (1999) showed that free ATP was also capable of sensitising C-MH fibres to heat. In the same study they found that only a small number of nociceptors tested were activated by low concentrations of ATP (less than 1 m m) but the results were not reported in detail (). In the present study we mostly used,β-methylene ATP, an ATP analogue that is more stable than free ATP and that may be more selective for P2X 1 and P2X 3 receptors (;;; ). We did not examine whether this agonist can also sensitise C-MH fibres to heat although it was clear that C-MH fibres were activated more robustly by this agonist than heat-insensitive C-fibres ( and ). Other groups have examined the effects of P2X agonists on primary afferents innervating visceral and joint tissues (Dow; ). The responses observed were sensitive to the non-specific P2X antagonist pyridoxal-phosphate-6-azophenyl-2′,4′-disulphonic acid (PPADS). In both studies no evidence was found that the effects of P2X agonists were selective for a particular afferent type as we show here for cutaneous receptors. • • Comparison of primary afferent response and nocifensive behaviour Data obtained from the present study are compared with already published data on the time course of nocifensive responses in behaving rats and humans (see text for details). The time course of the response to ATP analogues is shown in three different models: rat nocifensive behaviour following intraplantar injection of,β-methylene ATP (▵, dashed line) (), transcutaneous iontophoresis of ATP in humans (□, continuous line) () and single C-fibre unit recordings from the in vitro skin-nerve preparation (• with error bars, continuous line). The method of quantifying the response to ATP analogues is indicated in the y-axis: paw lifting in 2 min bins for the rat behavioural model; the average number of spikes in 10 s bins for the single unit recordings; and the average visual analog score (VAS rating) every 20 s for human subjects. Recent results from mice lacking P2X 3 receptors indicate that this ATP-gated channel is responsible for a large proportion of rapidly desensitising ATP responses observed in sensory neurons (; ). It has been known for some time that P2X 3 receptors are predominantly localised on isolectin-B 4-positive nociceptive neurons (; ). These neurons terminate in a different region of the spinal cord than do IB 4-negative sensory neurons and have distinctive neurotrophic requirements (Bradbury et al.1998; Snider & McMahon, 1998). In addition they have distinctive membrane properties and they may possess more limited regenerative capacity than IB 4-negative nociceptive neurons (; ). Here we show that noxious heat-sensitive C-fibre nociceptors (C-MH fibres) have a much more robust response to the P2X agonist,β-methylene ATP than do C-mechanonociceptors. Around half of the isolectin B 4-positive sensory neurons do indeed respond to noxious heat (). Thus at the receptor terminals it is likely that neurons responsive to noxious heat and to P2X agonists are IB 4 positive. However, ATP can still evoke some nocifensive responses in mice lacking the P2X 3 receptor (), and thus the activation of sensory neurons by ATP or its analogue cannot be taken definitively to mean that P2X 3 receptors are involved. Instead it must be assumed that a proportion of the nociceptor response we observed here to,β-methylene ATP was due to the activation of yet uncharacterised purinergic receptors. In earlier studies using isolated neurons it was found that sensory neurons with nociceptive properties possessed ATP-gated currents distinct from those of non-nociceptive neurons (Coo). Thus a large rapidly desensitising current appeared to be specific to nociceptors and was attributed to the presence of P2X 3 homomultimers. Recent work with mice lacking the P2X 3 receptor has confirmed that P2X 3 is necessary for the large rapidly desensitising current in presumptive nociceptive sensory neurons (; ). Behavioural experiments also showed that these P2X 3-deficient mice have a reduced nocifensive response to the injection of ATP into the paw. These nocifensive responses normally have the same time course as the activation of nociceptors that we have described here (). This is surprising as it suggests that the native P2X 3 receptors activated by,β-methylene ATP and ATP in the periphery have quite different inactivation kinetics from that in the cell soma. In the periphery action potentials were generated in the presence of the agonist during the entire course of a 5 min application. In contrast in the cell soma of tooth pulp nociceptors the sustained current evoked by,β-methylene ATP is very small and inactivates with a time course of hundreds of milliseconds (Coo). Interestingly, the same cells could respond to ATP with a train of action potentials maintained over several seconds when recorded in current clamp mode (see Coo). It is conceivable that ATP sequentially activates P2X 3 channels that are spatially separated from each other within the receptive field. The receptive field of nociceptive C-fibres can be 2-3 mm in diameter and as such the membrane surface exposed to ATP is several orders of magnitude larger than the cell soma. Action potentials might be initiated at different points within the receptive field but the initiation zone for each would show desensitisation as in the soma. We did, however, observe that the response during continued agonist application could decrease by up to 50% during the stimulus (). This pattern of response correlates well with the time course of the lifting response elicited by ATP analogues in rats (Bland-Ward & Humphrey, 1997; ) and the desensitisation of the pain response elicited in humans () (). What is the physiological function of P2X receptors on nociceptive neurons? It has been proposed that during tissue inflammation or damage increased concentrations of extracellular ATP could activate nociceptors and thus contribute to hyperalgesia (Burnstoc). Experiments with mice deficient in P2X 3 demonstrate that activation of these receptors in the periphery contributes to pain behaviours as P2X 3-deficient mice exhibit less formalin-induced pain than control mice (; ). It is of interest that the dose of ATP required to produce pain in humans or nocifensive responses in rodents is reduced dramatically (100 times lower) after inflammation (). In this study we have demonstrated that this potentiation is explained by a large increase in the response of individual nociceptors to P2X agonists () combined with an increase in the number of nociceptors responding to low concentrations of ATP. If one calculates the total afferent discharge to 50 μ m of,β-methylene ATP (mean discharge of all C-fibres including non-responders) then C-fibres innervating inflamed skin discharged on average 1.24 ± 0.29 spikes (2 min) −1 compared with 0.59 ± 0.28 spikes (2 min) −1 (Mann-Whitney U test, P. ATP activated channels in rat and bullfrog sensory neurons: concentration dependence and kinetics. Journal of Neuroscience 10, 1– 10. •| • • Belyantseva, I. Stability and plasticity of primary afferent projections following nerve regeneration and central degeneration. European Journal of Neuroscience 11, 457– 469. •| •| •| • • Bland-Ward, P. & Humphrey, P. Acute nociception mediated by hindpaw P2X receptor activation in the rat. British Journal of Pharmacology 122, 365– 371. •| •| •| • • Bleehen, T. Observations on the algogenic actions of adenosine compounds on the human blister base preparation. Pain 3, 367– 377. •| •| •| • • Bradbury, E. J., Burnstock, G. & McMahon, S. The expression of P2X 3 purinoreceptors in sensory neurons: effects of axotomy and glial-derived neurotrophic factor. Molecular and Cellular Neurosciences 12, 256– 268. •| •| •| • • Burnstock, G., McMahon, S. B., Humphrey, P. & Hamilton, S. ATP (P2X) receptors and pain. Proceedings of the 12th World Congress on Pain, Progress in Pain Research and Management, vol. IASP Press, Vienna • Chen, C. C., Akopian, A. N., Sivilotti, L., Colquuhoun, D., Burnstock, G. A P2X purinoreceptor expressed by a subset of sensory neurons. Nature 377, 428– 431. •| •| •| •| • • Cockayne, D. A., Hamilton, S. G., Zhu, Q.-M., Dunn, P. M., Zhong, Y., Novakovic, S., Malmberg, A. B., Cain, G., Berson, A., Kasssotakis, L., Hedley, L. R., Lachnit, W. G., Burnstock, G., McMahon, S. Urinary bladder hyporeflexia and reduced pain-behaviour in P2X 3-deficient mice. Nature 407, 1011– 1015. •| •| •| •| • • Collo, G., North, R. A., Kawashima, E., Merlo-Pich, E., Neidhart, S., Surprenant, A. Cloning of P2X5 and P2X6 receptors and the distribution and properties of an extended family of ATP-gated ion channels. Journal of Neuroscience 16, 2495– 2507. •| • • Cook, S. P., Vulchanova, L., Hargreaves, K. & McCleskey, E. Distinct ATP receptors on pain-sensing and stretch-sensing neurons. Nature 387, 505– 508. •| •| •| •| • • Coutts, A. A., Jorizzo, J. J., Greaves, M. & Burnstock, G. Adenosine triphosphate-evoked vascular changes in human skin: mechanism of action. European Journal of Pharmacology 76, 391– 401. •| •| •| • • Dowd, E., McQueen, D. S., Chessell, I. & Humphrey, P. P2X receptor-mediated excitation of nociceptive afferents in the normal and arthritic rat knee joint. British Journal of Pharmacology 125, 341– 346. •| •| •| • • Grubb, B. Characterization of cultured dorsal root ganglion neuron P2X receptors. European Journal of Neuroscience 11, 149– 154. •| •| •| • • Hamilton, S. & McMahon, S. The effects of inflammation and inflammatory mediators on nociceptive behaviour induced by ATP analogues in the rat. British Journal of Pharmacology 126, 326– 332. •| •| •| • • Hamilton, S. G., Warburton, J., Bhattacharjee, A., Ward, J. & McMahon, S. ATP in human skin elicits a dose related pain response which is potentiated under conditions of hyperalgesia. Brain 123, 1238– 1246. •| •| • • Kirkup, A. J., Booth, C. E., Chessell, I. P., Humphrey, P. Excitatory effect of P2X receptor activation on mesenteric afferent nerves in the anaesthetised rat. Journal of Physiology 520, 551– 563. •| •| •| • • Kocher, L., Anton, F., Reeh, P. & Handwerker, H. The effect of carrageenan-induced inflammation on the sensitivity of unmyelinated skin nociceptors in the rat. Pain 29, 363– 373. •| •| •| • • Koltzenburg, M., Bennett, D. L., Shelton, D. & McMahon, S. Neutralization of endogenous NGF prevents the sensitization of nociceptors supplying inflamed skin. European Journal of Neuroscience 11, 1698– 1704. •| •| •| • • Koltzenburg, M., Stucky, C. Receptive properties of mouse sensory neurons innervating hairy skin. Journal of Neurophysiology 78, 1841– 1850. •| • • Kress, M. & Guenther, S. Role of [Ca 2+] i in the ATP-induced heat sensitization process of rat nociceptive neurons. Journal of Neurophysiology 81, 2612– 2619. •| • • Kress, M., Koltzenburg, M., Reeh, P. & Handwerker, H. Responsiveness and functional attributes of electrically localized terminals of cutaneous C-fibres in vivo and in vitro. Journal of Neurophysiology 68, 581– 595. •| • • Lewis, C., Neidhart, S., Holy, C., North, R. A., Buell, G. & Surprenant, A. Coexpression of P2X2 and P2X3 receptor subunits can account for ATP-gated currents in sensory neurons. Nature 377, 432– 435. •| •| •| •| • • Petruska, J. C., Napaporn, J., Johnson, R. Subclassified acutely dissociated cells of rat DRG: histochemistry and patterns of capsaicin-, proton-, and ATP-activated currents. Journal of Neurophysiology 84, 2365– 2379. •| • • Reeh, P. Sensory receptors in mammalian skin in an in vitro preparation. Neuroscience Letters 66, 141– 146. •| •| •| • • Rongen, G. A., Floras, J. S., Lenders, J. W., Thien, T. Cardiovascular pharmacology of purines. Clinical Science 92, 13– 24. •| •| •| • • Ryan, L. M., Rachow, J. & McCarty, D. Synovial fluid ATP: a potential substrate for the production of inorganic pyrophosphate. Journal of Rheumatology 18, 716– 720. •| • • Snider, W. & McMahon, S. Tackling pain at the source: new ideas about nociceptors. Neuron 20, 629– 632. •| •| •| • • Souslova, V., Cesare, P., Ding, Y., Akopian, A. N., Stanfa, L. C., Suzuki, R., Carpenter, K., Dickenson, A., Boyce, S., Hill, R., Nebenius-Oosthuizen, D., Smith, A. Warm current deficits and aberrant inflammatory pain in mice lacking P2X 3 receptors. Nature 407, 1015– 1017. •| •| •| •| • • Stucky, C. Isolectin B(4)-positive and -negative nociceptors are functionally distinct. Journal of Neuroscience 19, 6497– 6505. •| • • Valera, S., Hussy, N., Evans, R. J., Adami, N., North, R. A., Surprenant, A. A new class of ligand-gated ion channel defined by P2x receptor for extracellular ATP. Nature 371, 516– 519. •| •| •| •| • • Vulchanova, L., Riedl, M. S., Shuster, S. J., Stone, L. S., Hargreaves, K. M., Buell, G., Surprenant, A., North, R. P2X3 is expressed by DRG neurons that terminate in inner lamina II. European Journal of Neuroscience 10, 3470– 3478. •| •| •| • • Zimmermann, H. Signalling via ATP in the nervous system. Trends in Neurosciences 17, 420– 426. •| •| •| • Related content. The role of protease activated receptor-2 (PAR-2) activation in trigeminal nociception and in induction of functional competence in the delta opioid receptor (DOR) is not known. In this study, we evaluated whether agonists of PAR-2 activate the capsaicin-sensitive subclass of trigeminal nociceptors in a PLC-PKC-dependent manner and induce functional competence in the DOR. Adult male rat trigeminal ganglion (TG) cultured neurons were treated with the PAR-2 agonist (SL-NH 2) or an enzyme activator of PAR (trypsin) and the activation of TG nociceptors was assessed using three independent methods: neuropeptide release, calcium influx, and whole cell patch-clamp. The specificity of SL-NH 2 and trypsin responses was evaluated using TG cultures transfected with siRNA against PAR-2. The in vivo role of PAR-2 activation was determined measuring SL-NH 2 and trypsin-evoked nocifensive behavior and increase in blood flow. Trigeminal neurons were treated with SL-NH 2/vehicle and then the DOR agonist to determine DOR inhibition of evoked neuropeptide release and cAMP accumulation. The results showed that SL-NH 2 (100 μM) and trypsin (1-600 nM) activate TG nociceptors, which is partly reversible by the PKC inhibitor bisindolylmaleimide (500 nM) and by ruthenium red (10 μM). In cultures treated with siRNA against PAR-2, both SL-NH 2 and trypsin responses were significantly diminished. Both SL-NH 2 and trypsin evoke nocifensive behavior and increases in blood flow in an orofacial pain model. Application of SL-NH 2 rapidly produced functional competence of DOR for inhibiting nociceptor function. In inflamed tissue, endogenous proteases may activate TG nociceptors and generate pain. Moreover, activation of PAR-2 can also induce functional competence in DOR. Abstract = 'The role of protease activated receptor-2 (PAR-2) activation in trigeminal nociception and in induction of functional competence in the delta opioid receptor (DOR) is not known. In this study, we evaluated whether agonists of PAR-2 activate the capsaicin-sensitive subclass of trigeminal nociceptors in a PLC-PKC-dependent manner and induce functional competence in the DOR. Adult male rat trigeminal ganglion (TG) cultured neurons were treated with the PAR-2 agonist (SL-NH2) or an enzyme activator of PAR (trypsin) and the activation of TG nociceptors was assessed using three independent methods: neuropeptide release, calcium influx, and whole cell patch-clamp. The specificity of SL-NH2 and trypsin responses was evaluated using TG cultures transfected with siRNA against PAR-2. The in vivo role of PAR-2 activation was determined measuring SL-NH2 and trypsin-evoked nocifensive behavior and increase in blood flow. Trigeminal neurons were treated with SL-NH2/vehicle and then the DOR agonist to determine DOR inhibition of evoked neuropeptide release and cAMP accumulation. The results showed that SL-NH2 (100 μM) and trypsin (1-600 nM) activate TG nociceptors, which is partly reversible by the PKC inhibitor bisindolylmaleimide (500 nM) and by ruthenium red (10 μM). In cultures treated with siRNA against PAR-2, both SL-NH2 and trypsin responses were significantly diminished. Both SL-NH2 and trypsin evoke nocifensive behavior and increases in blood flow in an orofacial pain model. Application of SL-NH2 rapidly produced functional competence of DOR for inhibiting nociceptor function. In inflamed tissue, endogenous proteases may activate TG nociceptors and generate pain. Moreover, activation of PAR-2 can also induce functional competence in DOR.' TY - JOUR T1 - PAR-2 agonists activate trigeminal nociceptors and induce functional competence in the delta opioid receptor AU - Patwardhan,Amol M. AU - Diogenes,Anibal AU - Berg,Kelly A. AU - Fehrenbacher,Jill C. AU - Clarke,William P. AU - Akopian,Armen N. AU - Hargreaves,Kenneth M. PY - 2006/11 Y1 - 2006/11 N2 - The role of protease activated receptor-2 (PAR-2) activation in trigeminal nociception and in induction of functional competence in the delta opioid receptor (DOR) is not known. In this study, we evaluated whether agonists of PAR-2 activate the capsaicin-sensitive subclass of trigeminal nociceptors in a PLC-PKC-dependent manner and induce functional competence in the DOR. Adult male rat trigeminal ganglion (TG) cultured neurons were treated with the PAR-2 agonist (SL-NH2) or an enzyme activator of PAR (trypsin) and the activation of TG nociceptors was assessed using three independent methods: neuropeptide release, calcium influx, and whole cell patch-clamp. The specificity of SL-NH2 and trypsin responses was evaluated using TG cultures transfected with siRNA against PAR-2. The in vivo role of PAR-2 activation was determined measuring SL-NH2 and trypsin-evoked nocifensive behavior and increase in blood flow. Trigeminal neurons were treated with SL-NH2/vehicle and then the DOR agonist to determine DOR inhibition of evoked neuropeptide release and cAMP accumulation. The results showed that SL-NH2 (100 μM) and trypsin (1-600 nM) activate TG nociceptors, which is partly reversible by the PKC inhibitor bisindolylmaleimide (500 nM) and by ruthenium red (10 μM). In cultures treated with siRNA against PAR-2, both SL-NH2 and trypsin responses were significantly diminished. Both SL-NH2 and trypsin evoke nocifensive behavior and increases in blood flow in an orofacial pain model. Application of SL-NH2 rapidly produced functional competence of DOR for inhibiting nociceptor function. In inflamed tissue, endogenous proteases may activate TG nociceptors and generate pain. Moreover, activation of PAR-2 can also induce functional competence in DOR. AB - The role of protease activated receptor-2 (PAR-2) activation in trigeminal nociception and in induction of functional competence in the delta opioid receptor (DOR) is not known. In this study, we evaluated whether agonists of PAR-2 activate the capsaicin-sensitive subclass of trigeminal nociceptors in a PLC-PKC-dependent manner and induce functional competence in the DOR. Adult male rat trigeminal ganglion (TG) cultured neurons were treated with the PAR-2 agonist (SL-NH2) or an enzyme activator of PAR (trypsin) and the activation of TG nociceptors was assessed using three independent methods: neuropeptide release, calcium influx, and whole cell patch-clamp. The specificity of SL-NH2 and trypsin responses was evaluated using TG cultures transfected with siRNA against PAR-2. The in vivo role of PAR-2 activation was determined measuring SL-NH2 and trypsin-evoked nocifensive behavior and increase in blood flow. Trigeminal neurons were treated with SL-NH2/vehicle and then the DOR agonist to determine DOR inhibition of evoked neuropeptide release and cAMP accumulation. The results showed that SL-NH2 (100 μM) and trypsin (1-600 nM) activate TG nociceptors, which is partly reversible by the PKC inhibitor bisindolylmaleimide (500 nM) and by ruthenium red (10 μM). In cultures treated with siRNA against PAR-2, both SL-NH2 and trypsin responses were significantly diminished. Both SL-NH2 and trypsin evoke nocifensive behavior and increases in blood flow in an orofacial pain model. Application of SL-NH2 rapidly produced functional competence of DOR for inhibiting nociceptor function. In inflamed tissue, endogenous proteases may activate TG nociceptors and generate pain. Moreover, activation of PAR-2 can also induce functional competence in DOR. KW - Delta opioid receptor KW - Pain KW - PAR, Protease activated receptor KW - RNAi KW - siRNA KW - Trigeminal KW - Trypsin UR - UR - U2 - 10.1016/j.pain.2006.05.007 DO - 10.1016/j.pain.2006.05.007 M3 - Article VL - 125 SP - 114 EP - 124 JO - Pain T2 - Pain JF - Pain SN - 0304-3959 IS - 1-2 ER.  
If you like to get most of your news on the web, then you've likely struggled with reading things on your iPhone or iPad, since ads and menus can clutter up the page. Reader View in Safari allows you to view web pages without all that clutter and you can even change the font, font size, and page color to better suit your reading habits and vision. Here's how to use Reader View in Safari on iPhone and iPad. • • • How to enable Reader View When you're on a website that supports Reader View on your iPhone or iPad, the address bar at the top of the screen will say 'Reader View Available' when you arrive at the site. If it's not there, then it can't be used. • Launch Safari from your Home screen. • Navigate to the website you'd like to read. • Tap the Reader button on the left of the address bar. It looks like a series of stacked lines. Reader View is now enabled. You'll notice that much of the color and animations will be removed and you'll see a simple screen of text. To disable Reader View, just tap the Reader button again. How to change the font and font size in Reader View • Launch Safari from your Home screen. • Navigate to the website you'd like to read. How to Automatically Open Articles in Safari. For example, you could enable Reader Mode for all websites and then tell Safari not to use Reader Mode. Want to visit websites without annoying ads, videos, and other distractions? Safari's Reader mode is the way to go. Does anyone know a WordPress theme that does NOT allow Safari reader mode? Offerings — as I find Safari’s Reader a very. To enable Safari Reader on. 
Oct 12, 2017 iOS 11 and macOS High Sierra come with a little-known shortcut in Safari that lets you automatically invoke Reader mode for specific websites. We show you how. By Joe Hutsko, Barbara Boyd. If Safari perceives a readable article on the page you are viewing, the Reader button is active in the Search and Address field, and you have the option to open the article in Reader. Safari aims to be elegant and clutter-free, so Reader removes all the ads, buttons, bells, and whistles from the. • Tap the Reader button on the left of the address bar. It looks like a series of stacked lines. • Tap the A's on the right of the address bar. • Tap either of the A's that appear in the menu to change the font to one of two sizes. • Tap one of the eight font choices. How to change the page color in Reader View If you reader better when text is light and the page is dark, you can change things to your liking. • Launch Safari from your Home screen. • Navigate to the website you'd like to read. • Tap the Reader button on the left of the address bar. It looks like a series of stacked lines. • Tap the A's on the right of the address bar. • Tap one of the four page color choices: white, beige, gray, or black. Let us know in the comments below. Apple Footer This site contains user submitted content, comments and opinions and is for informational purposes only. Apple may provide or recommend responses as a possible solution based on the information provided; every potential issue may involve several factors not detailed in the conversations captured in an electronic forum and Apple can therefore provide no guarantee as to the efficacy of any proposed solutions on the community forums. Apple disclaims any and all liability for the acts, omissions and conduct of any third parties in connection with or related to your use of the site. All postings and use of the content on this site are subject to the. Despite being one of the most pirated software vendors in the world, Microsoft doesn’t have a long track record of cracking down on individual pirates. In fact, two months ago Microsoft CEO Satya Nadella noted that in some cases piracy can act as a conversion tool. “We’ve always had freemium. Sometimes our freemium was called piracy,” Nadella, adding that the usage first approach has its advantages. This doesn’t mean that all pirates can have their way though. Microsoft does keep a close eye on the unauthorized use of its products with help from its in house cybercrime center. Late last week Microsoft filed a copyright infringement lawsuit against a person (or persons) who activated pirated copies of Windows 7 and Office 10 from an AT&T Internet connection. “Microsoft’s cyberforensics have identified a number of product key activations originating from IP address 76.245.7.147, which is presently assigned to ISP AT&T Internet Services.,” the complaints () reads. “These activations have characteristics that on information and belief, establish that Defendants are using the IP address to activate pirated software.” While many people believe that unauthorized copies are hard for Microsoft to detect, the company explains that its cybercrime team leverages state-of-the-art technology to detect software piracy. The company describes its investigative approach as cyberforensics. Among other things, they look for activation patterns and characteristics which make it likely that certain IP-addresses are engaged in unauthorized copying. “As part of its cyberforensic methods, Microsoft analyzes product key activation data voluntarily provided by users when they activate Microsoft software, including the IP address from which a given product key is activated,” the company writes. According to the complaint, the defendant(s) in this case have activated numerous copies of Windows 7 and Office 2010 with suspicious keys. These keys were likely stolen from Microsoft’s supply chain, used without permission from the refurbisher channel, and used more often than the license permits. Microsoft is now looking to identify the person or persons responsible for the copyright and trademark infringements, to recoup the damage they’ve suffered. From the descriptions used in the complaint it seems likely that the target is not an average user, but someone who sells computers containing pirated software. Time will tell whether that’s indeed the case.  
Windows 10 upgrades from pirated predecessors won't be legit. You can reset it, but it will continue to turn black until you activate Windows. This is a list of the Gold Bolt locations for Ratchet& Clank Future: Tools of Destruction. A page for describing ParanoiaFuel: Video Games. The following works have their own pages: Overwatch Watch_Dogs Minecraft has Ghasts, whose cries are.  
PROTECT THIS CARD LIKE CASH. Redeemable for merchandise or services (excluding gift cards) in JCPenney Stores in the U.S. Or PR and jcp.com. Not redeemable for cash except where required by law. May not be used to pay on credit accounts. Lost, stolen or damaged gift cards may be cancelled and replaced with a new gift card in the amount of the then remaining balance upon proof as required by JCPenney. Contact your local JCPenney Store for details. 
Card has no value until activated. No fees or expiration date. For Balance Inquiry or Customer Service, call 1-800-294-8804 or for Balance Inquiry visit www.jcp.com. All trademarks are property of their respective owners. © 2015 Blackhawk Network, Inc. All Rights Reserved. 
Designated trademarks and brands are the property of their respective owners. Use of this website constitutes acceptance of GiftCardLab's and *Visa ® Gift Cards may be used wherever Visa debit cards are accepted in the US. No cash access. Visa Gift Cards and Visa Incentive Cards are issued by MetaBank ®, Member FDIC, pursuant to a license from Visa U.S.A. The 5% Back Bonus offer is provided by the Award Sponsor and is not provided or endorsed by MetaBank or Visa. I had the card for a while and lost the the phone number you call to activate the card. Jcpenney Credit Card Phone Number. Call the customer service number that is printed on the back of the card and tell them that you want to activate it and lost the strip with the activation telephone number. Your JCPenney Credit Card (back to top) Do I need to call to activate my card before I use it? You may need to call to activate your card. Home: Apply now: My account: Pay my bill: JCPenney Rewards: Card benefits: Gold & Platinum status: FAQs. JCP Credit Today Activate a JCP credit card and save 20% on a purchase Returning customers can sign in to pay a bill, view recent transactions and update. Get notified of changes to your TransUnion® report with 100% free CreditWise® from Capital One. Are you eligible for pre-qualified credit card offers? 
Many Handsets have Advance Mobile Tracker facility. Advance Mobile tracker is a security feature that helps recover lost or stolen phones. An updated version, Advance Mobile tracker improves its services by securing information and phone calls to your mobile even after it is lost or stolen. Owners can remotely remove all the data from the missing phone, so that none of the information stored on the mobile can be accessed by other people. In addition, calls can be redirected to another handset, ensuring you won’t miss a call even though you have lost your mobile. The feature will also alert you to the general location of your mobile, helping you recover lost or misplaced phones. Use these steps to support voice commands on the Samsung Galaxy S 5. On this page: Open an app Voice dial a contact Write a text message (SMS) with. Find device-specific support and online tools for your Samsung Galaxy J1. Popular Content. Top 10 Things to Do with Your Smartphone Block Numbers Overview Device Replacement Program Transfer Contacts & Other Content Between Devices. Device-Specific Support. View your User Guide (PDF) Learn how to use.  
• In Menu mode, select Settings → Security → Mobile tracker 2.0 → Settings. • Enter a new 8-digit password for the mobile tracker and press the Confirm key. • Enter the new password again and press the Confirm key. • Scroll left or right to On. • Scroll down and press → Add recipient to enter the recipient list. • Press → Contacts to open your contact list. • Scroll to a contact and press. • Select a number (if necessary). • Press → Ok to save the recipient list. • Scroll down and press → Edit to enter the sender’s name. • Enter the sender’s name and press the Confirm key. • Press → Save. • Press → Yes. To change the password for the mobile tracker, select Change password. Samsung included a new 'SOS' feature on the Galaxy S6 and S6 Edge that could literally be a lifesaver should you ever find yourself in an emergency situation. It's one of those things that we hope to never have to use, but will be very thankful for if a need ever arises. The feature can be activated in an emergency situation with three quick taps of your power button, and it will immediately send pictures from your front and rear camera to a set of emergency contacts. In addition to that, it sends a message stating that you need help, with your exact location and an audio recording. But SOS must be set up before it can be used, so what better time than right now? Step 3: Using the SOS Feature For this feature to work properly, you'll need to be using the stock Messaging app that came pre-installed on your Galaxy S6. In other words, if you've set a third-party messaging app as your default SMS client, the SOS feature will not work. In the event of an emergency, simply tap your Galaxy's power button three times in rapid succession—it does not matter if your screen is on or off for this to work. At this point, you'll see an ongoing notification informing you that SOS mode was activated. 
Here is a quick and easy video to show you how to perform a print screen / screenshot on a Windows laptop. It's really easy to get started and create your first screenshot. All you have to do is get something on the screen you want to take a screenshot/screen dump of and press the following key (can change per machine). PRTSC PRTSCN Once this key has been pressed it will take a snapshot of the current screen and put it in the copy and paste buffer. You can then 'paste' this screen shot in lots of different applications such as graphic and photo software and writing packages. To 'paste' the image in the application (MS Word, Paint etc) you then visit the 'Edit' menu and select 'Paste'. 
This will then put the screenshot in the document. How to take a computer screen shot How to use printscreen How to print screen on Windows 7 How to print screen on Windows 8 How to print screen on laptop How to take screen shots on a laptop How to print screen laptop screenshot. Alternative for Print screen Button on Windows 10 in the case as Keyboard does not function efficiently or confront mechanical disorder is recommended here. 
|
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2018
Categories |
 RSS Feed
RSS Feed
